A stoichiometric mixture of ethane and air reacts at 15C. Using the following values for Cp...
Fantastic news! We've Found the answer you've been seeking!
Question:
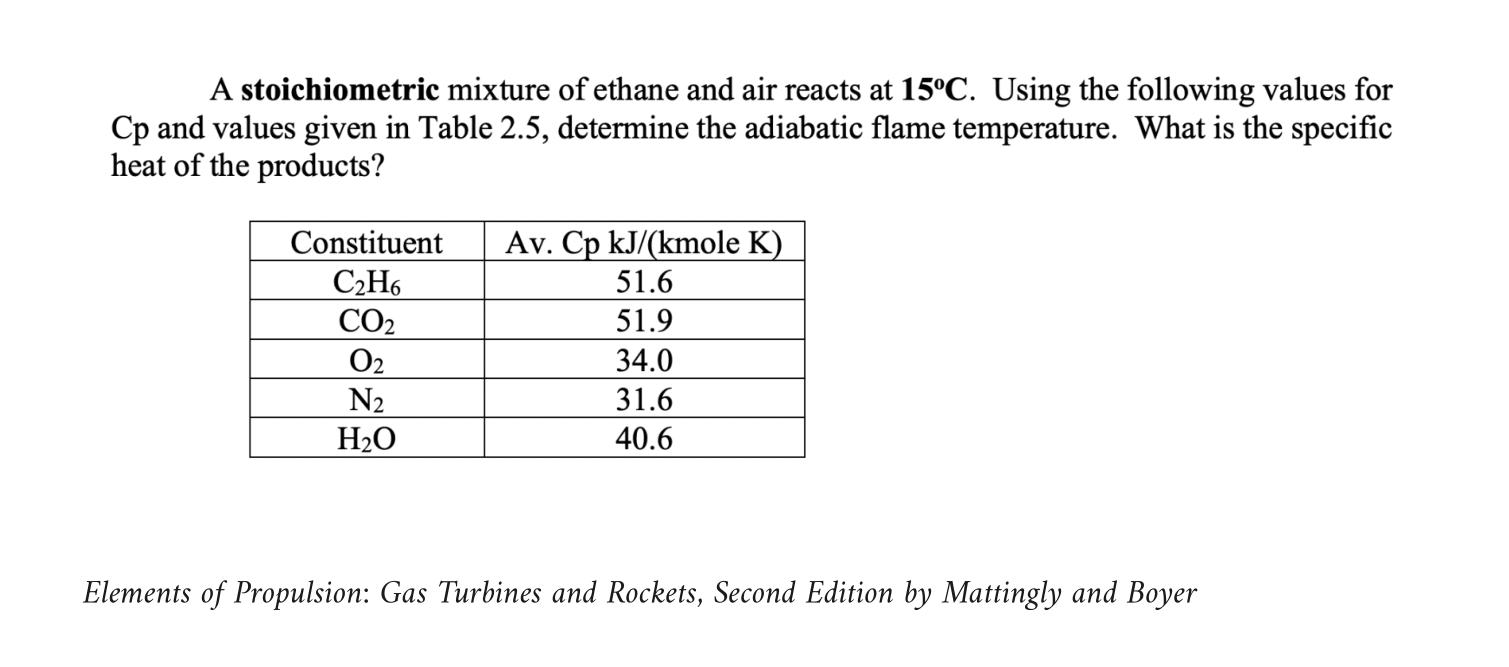
Transcribed Image Text:
A stoichiometric mixture of ethane and air reacts at 15°C. Using the following values for Cp and values given in Table 2.5, determine the adiabatic flame temperature. What is the specific heat of the products? Constituent C₂H6 CO₂ 0₂ N₂ H₂O Av. Cp kJ/(kmole K) 51.6 51.9 34.0 31.6 40.6 Elements of Propulsion: Gas Turbines and Rockets, Second Edition by Mattingly and Boyer A stoichiometric mixture of ethane and air reacts at 15°C. Using the following values for Cp and values given in Table 2.5, determine the adiabatic flame temperature. What is the specific heat of the products? Constituent C₂H6 CO₂ 0₂ N₂ H₂O Av. Cp kJ/(kmole K) 51.6 51.9 34.0 31.6 40.6 Elements of Propulsion: Gas Turbines and Rockets, Second Edition by Mattingly and Boyer
Expert Answer:
Answer rating: 100% (QA)
To determine the adiabatic flame temperature and the specific heat of the products we need to use the given values for Cp and perform some calculations First lets calculate the heat of reaction for th... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, ? Ronald W. Rousseau, ? Lisa G. Bullard
Posted Date:
Students also viewed these finance questions
-
Write a 2-page report to show the impact on the GDP, unemployment and inflation in Canada right after the pandemic hit in March 2020. First, analyze the situation using the AS-AD model. Then research...
-
what entrepreneurial habits did harish hande exhibit in hadling SELCO? If you're hande's position, would you do the same? Why or why not? See attached picture for reference. ELMS REVIEW Harish Hande...
-
A stoichiometric mixture of benzene, C6H6, and air is mixed from the reactants flowing at 25C, 100 kPa. Find the adiabatic flame temperature. What is the error if constant specific heat at T0...
-
In Exercises 7683, use a graphing utility to graph the function. Use the graph to determine whether the function has an inverse that is a function (that is, whether the function is one-to-one). f(x)...
-
The following statements can be found in the IRS instructions for Form 1040. Give the inverse, converse, contra-positive, and negation of each. Simplify and use De Morgan's laws when possible. (a) If...
-
A rectangular cast-iron column 16.0 cm x 16.0 cm x 4.50 m supports a weight of 7.95 x 106 N. Find the stress on the top of the column.
-
For an endothermic reaction, \(H\) is (a) - ve (b) 0 (c) + ve (d) Unpredictable.
-
The IRS is concerned with improving the accuracy of tax Information given by its representatives over the telephone. Previous studies involved asking a set of 25 questions of a large number of IRS...
-
In water cooling tower air enters at a height of 1 m above the ground level and leaves at a height of 7 m. The inlet and outlet velocities are 20 m/s and 30 m/s respectively. Water enters at a height...
-
Reyes Rides is owned by Jason Reyes. The company has an August 31 fiscal year end and prepares adjustments on an annual basis. The following is an alphabetical list of its accounts at August 31,...
-
A. B. C. Give a detailed definition of "inventory", based on IAS2, Inventories. (6 marks) Use suitable examples to explain the following terms with regards to IAS 2, Inventories: i. Cost (3 marks) (3...
-
Many multinational and global companies use ethnocentric staffing in their operations abroad. Why do you think a company might prefer to have someone from the home country in top management...
-
Williamson Company, a furniture manufacturer, produces 10 000 units of Product X-100 each year for use in its production line. The costs per unit for Product X-100 are as follows: Direct materials:...
-
At the beginning of 2017, AB Corporation (a calendar-year corporation) owned the following assets: On February 1, 2017, AB sold its office furniture. On March 15, 2017, AB sold its computer...
-
Phonegrip Company manufactures mobile phone holders. Phonegrip currently buys a magnet for its phones for $0.90 per unit. Phonegrips president asked for cost estimates for making this product, and...
-
June and John decide to form a business. They each plan to contribute $20,000 in exchange for a 50 percent interest in the business. They will then take out a bank loan for $30,000 to cover the...
-
if you purchase a property with the business in 2016 for $310,000. In 2022 you want to sell that property with business. And if somebody gave you an offer for $605,000. That number is broken down...
-
In each of the following independent cases, document the system using whatever technique(s) your instructor specifies. a. Dreambox Creations (www.dreamboxcreations.com/) in Diamond Bar, California,...
-
The radiant energy incident on the earths surface on a sunny day is roughly 900 W/m2. Collecting and focusing sunlight and using the focused beam to heat a fluid is an old idea, and as the...
-
A steam trap is a device to purge steam condensate from a system without venting uncondensed steam. In one of the crudest trap types, the condensate collects and raises a float attached to a drain...
-
A natural gas is analyzed and found to consist of 85.5% v/v (volume percent) methane, 8.5% ethane, 2.5% propane, and 3.5% N 2 (noncombustible). (a) Calculate the higher and lower heating values of...
-
Frosting of evaporator tubes results in (a) increased heat transfer rate (b) decreased heat transfer rate (c) increased compressor pressure (d) decreased compressor pressure
-
The chemical formula of R-12 is (a) CCl2F (b) CClF3 (c) CCl2F2 (d) CHClF2
-
The use of chlorofluoro carbon refrigerants is banned because these react with (a) water vapours and cause acid rain (b) plants and cause green house effect (c) oxygen and cause its depletion (d)...

Study smarter with the SolutionInn App


