Question: A student performed three CHCl2 layer titrations in Part 2 using a standard sodium thiosulfate solution with a concentration of 0.01153 M. They reported
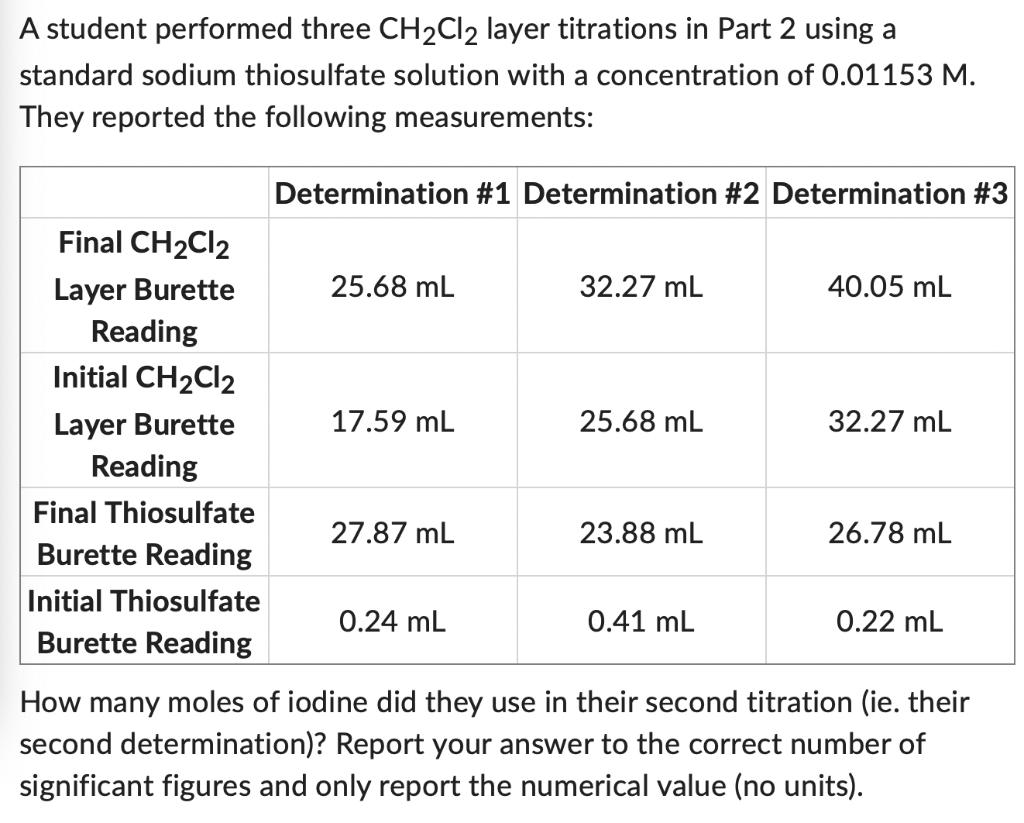
A student performed three CHCl2 layer titrations in Part 2 using a standard sodium thiosulfate solution with a concentration of 0.01153 M. They reported the following measurements: Final CHCl2 Layer Burette Reading Initial CHCl2 Layer Burette Reading Final Thiosulfate Burette Reading Initial Thiosulfate Burette Reading Determination #1 Determination #2 Determination #3 25.68 mL 17.59 mL 27.87 mL 0.24 mL 32.27 mL 25.68 mL 23.88 mL 0.41 mL 40.05 mL 32.27 mL 26.78 mL 0.22 mL How many moles of iodine did they use in their second titration (ie. their second determination)? Report your answer to the correct number of significant figures and only report the numerical value (no units).
Step by Step Solution
3.40 Rating (156 Votes )
There are 3 Steps involved in it
Step 11 The number of moles of iodine used in the second titration can be calculated using the formu... View full answer

Get step-by-step solutions from verified subject matter experts


