You want to prepare a solution with a concentration of 200.0 M from a stock solution...
Fantastic news! We've Found the answer you've been seeking!
Question:
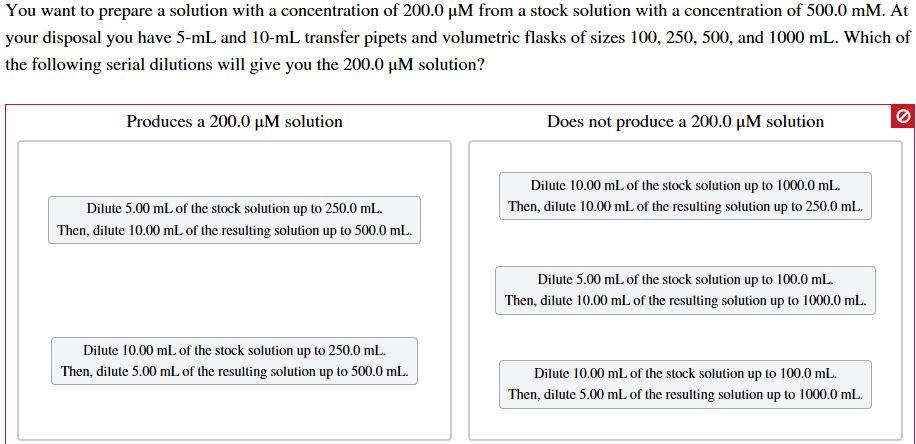
Transcribed Image Text:
You want to prepare a solution with a concentration of 200.0 µM from a stock solution with a concentration of 500.0 mM. At your disposal you have 5-mL and 10-mL transfer pipets and volumetric flasks of sizes 100, 250, 500, and 1000 mL. Which of the following serial dilutions will give you the 200.0 µM solution? Produces a 200.0 µM solution Does not produce a 200.0 µM solution Dilute 10.00 ml of the stock solution up to 1000.0 mL. Dilute 5.00 mL of the stock solution up to 250.0 mL. Then, dilute 10.00 ml of the resulting solution up to 250.0 ml. Then, dilute 10.00 mL of the resulting solution up to 500.0 mL. Dilute 5.00 ml of the stock solution up to 100.0 mL. Then, dilute 10.00 ml of the resulting solution up to 1000.0 ml. Dilute 10.00 mL of the stock solution up to 250.0 mL. Then, dilute 5.00 mL of the resulting solution up to 500.0 mL. Dilute 10.00 mL of the stock solution up to 100.0 mL. Then, dilute 5.00 mL of the resulting solution up to 1000.0 mL. You want to prepare a solution with a concentration of 200.0 µM from a stock solution with a concentration of 500.0 mM. At your disposal you have 5-mL and 10-mL transfer pipets and volumetric flasks of sizes 100, 250, 500, and 1000 mL. Which of the following serial dilutions will give you the 200.0 µM solution? Produces a 200.0 µM solution Does not produce a 200.0 µM solution Dilute 10.00 ml of the stock solution up to 1000.0 mL. Dilute 5.00 mL of the stock solution up to 250.0 mL. Then, dilute 10.00 ml of the resulting solution up to 250.0 ml. Then, dilute 10.00 mL of the resulting solution up to 500.0 mL. Dilute 5.00 ml of the stock solution up to 100.0 mL. Then, dilute 10.00 ml of the resulting solution up to 1000.0 ml. Dilute 10.00 mL of the stock solution up to 250.0 mL. Then, dilute 5.00 mL of the resulting solution up to 500.0 mL. Dilute 10.00 mL of the stock solution up to 100.0 mL. Then, dilute 5.00 mL of the resulting solution up to 1000.0 mL.
Expert Answer:
Answer rating: 100% (QA)
2 Answer 500 mm Given stock solution Concentration Required Concentration from stock 200 MM ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
You want to prepare a buffer solution that has a pH equal to the pKa of the acid component of the buffer. If you have 100 mL of a 0.10 M solution of the acid HA, what volume and concentration of NaA...
-
You want to prepare 500.0 mL of 1.000 M KNO3 at 20C, but the lab (and water) temperature is 24C at the time of preparation. How many grams of solid KNO3 (density 2.109 g/mL) should be dissolved in a...
-
The BSA standard solution with the concentration of 0.5 mg/mL was used to produce standard curve for protein assay based on Bradford method The test tubes contains BSA solutions was prepared: 0, 20,...
-
ARTICLE: Reference Resolve Asset Management's Yes, You Can Eat Sharpe Ratios. 1- The author spoke to two ways for the foundation to meet its target return of 5%. Identify what the two ways presented...
-
Do you think Microsoft should be broken up into two or even three separate corporations? Give at least two reasons to support your conclusion.
-
Access the March 26, 2015, filing of the 10-K report of Home Depot for the year ended February 1, 2015, from www.SEC.gov (ticker HD). Refer to Home Depot's balance sheet, including its note 4 (on...
-
What is a Mission System (Producer) role?
-
Lisa Surowsky was asked to help in determining the best ordering policy for a new product. Currently, the demand for the new product has been projected to be about 1,000 units annually. To get a...
-
Assume the following: The standard price per pound is $3.00. The actual quantity of materials purchased and used in production is 61,800 pounds. The actual purchase price per pound of materials was...
-
1. Based on the marketing activities DNBC has tried, do you think it has a production-oriented marketing or a consumer-oriented philosophy? Explain your reasoning with examples. 2. Research the City...
-
(c) As a software engineer, you have been tasked with establishing a software reuse programme within the software company where you work in order to improve the quality of the software produced by...
-
Explain what approach the efforts will take and explain why the strategy or tactic was considered for the desired outcome for the Latino Community Empowerment Center proposal.
-
Hi I would please need help with this questions - what Project Aristotle found to be most important for team effectiveness. - which is the most important of these factors, and WHY? - How will you use...
-
In a "Short-Paragraph", please respond to the following question,and include a "Source" or "Reference" at the end of your paragraph: Question: How are small businesses capable of providing additional...
-
How can the mining corporations should limit their usage of the FIFO workforce and finding other alternatives solutions ?
-
Do you think that unions have outlived their usefulness? Is there still a need for unions in the workplace?
-
Evaluating each market segment's attractiveness and selecting one or more segments to serve.
-
Why should you not model a decision variable as a random variable with a probability distribution?
-
What is the pH of a buffer solution that is 0.15 M chloroacetic acid and 0.10 M sodium chloroacetate? Ka = 1.3 10-3.
-
A sample of sodium thiosulfate, Na2S2O3, contains sulfur-35. Determine the mass of 35S in the sample from the decay rate, which was determined to be 7.7 1011 disintegrations per second. The...
-
If adenine, thymine, guanine, and cytosine were each analyzed separately in a sample of DNA, what molar ratios of A:T and G : C would you expect to find?
-
What are some reasons a potential prospect might not be readily accessible? How far should you go to try to overcome such an accessibility problem before you move to the next lead?
-
List three or four criteria you could use to qualify a lead as a likely prospect. How would you find out if the lead meets these criteria?
-
Who is currently in your own network that you could use for prospecting? How might you add to your network?

Study smarter with the SolutionInn App


