A student proposes the following Lewis structure for the dinitrogen monoxide (NO) molecule. N=0=N Assign a...
Fantastic news! We've Found the answer you've been seeking!
Question:
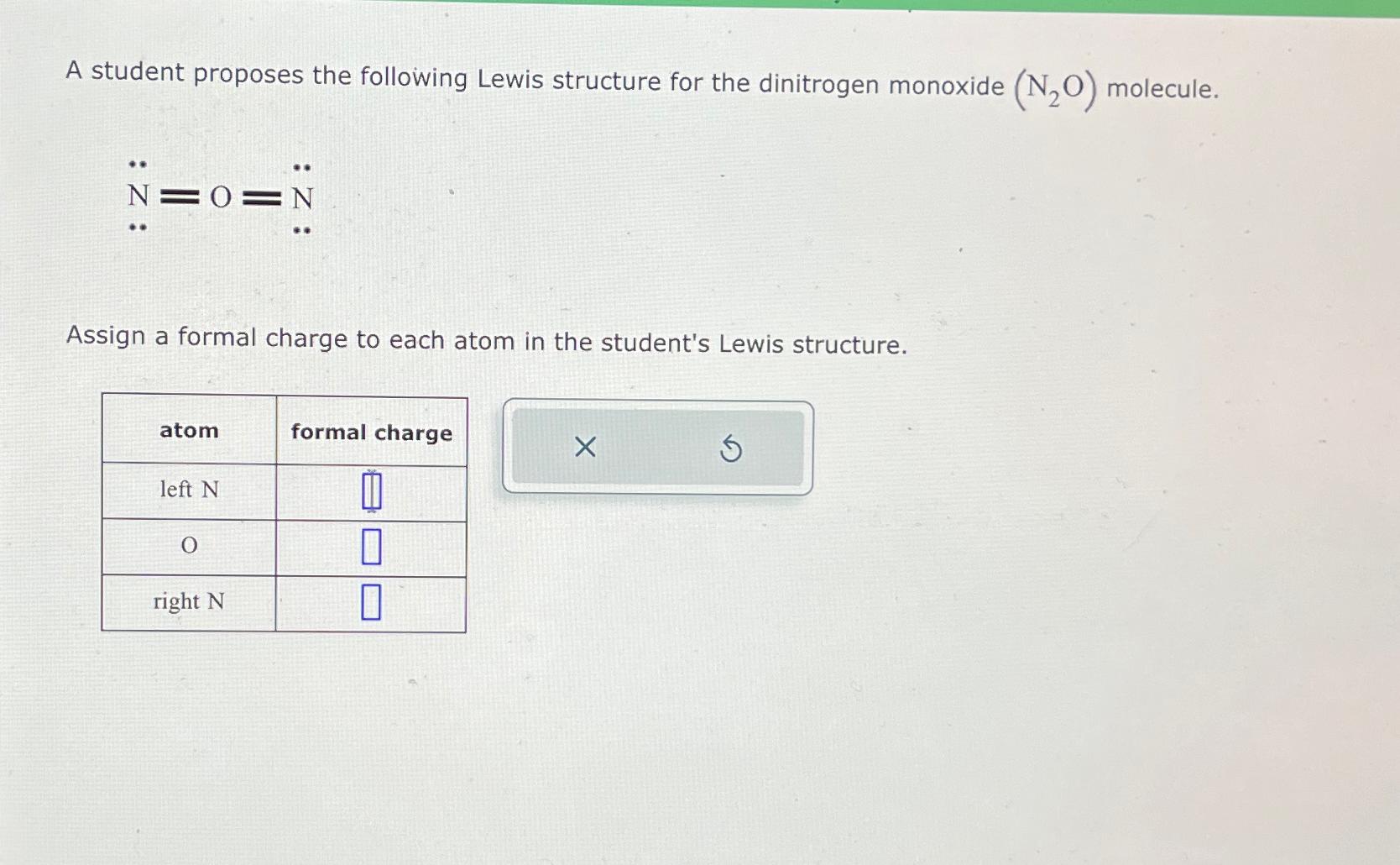
Transcribed Image Text:
A student proposes the following Lewis structure for the dinitrogen monoxide (NO) molecule. N=0=N Assign a formal charge to each atom in the student's Lewis structure. atom left N ** O right N formal charge 0 1 1 X A student proposes the following Lewis structure for the dinitrogen monoxide (NO) molecule. N=0=N Assign a formal charge to each atom in the student's Lewis structure. atom left N ** O right N formal charge 0 1 1 X
Expert Answer:
Answer rating: 100% (QA)
To calculate the formal charge of each atom in the Lewis structure for dinitrogen monoxide NO we can use the formula Formal charge Valence electrons Nonbonding electrons 05Bonding electrons Valence electrons are the number of electrons an atom has in its outer shell in the elemental state Nonbonding electrons are the electrons that an atom has in lone pairs in the Lewis structure Bonding electrons are the electrons shared between atoms in bonds From the Lewis structure provided The left nitrogen N atom has 1 lone pair 2 nonbonding electrons and is involved in a triple bond with the oxygen atom O sharing 6 bonding electrons The oxygen O atom has 2 lone pairs 4 nonbonding electrons and is involved in a triple bond with left nitrogen sharing 6 bonding electrons and a double bond with right nitrogen sharing 4 bonding electrons The right nitrogen N atom has 1 lone pair 2 nonbonding electrons and is involved in a double bond with the oxygen atom sharing 4 bonding electrons Lets calculate the formal charge for each atom 1 Left nitrogen N Valence electrons N ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The graph of a function f is given. Estimate 10 0 f(x) dx using five subintervals with (a) Right endpoints (b) Left endpoints (c) Midpoints. y FA 1 0 x
-
An airplane is flying at 32,000 feet when it starts its decent. It is descending at a rate of 2000 feet per minute. Let x represent the minutes of the flight's descent and y represent the planes...
-
According to Bradley, in order to succeed in the Digital Vortex, companies need to develop a meta-capability called "digital business agility". (a) Describe the three pillars of "digital business...
-
The function (x) = e x + x, being differentiable and one-to-one, has a differentiable inverse -1 (x). Find the value of d -1 /dx at the point (ln 2).
-
Two conspirators, Timothy Carpenter and Timothy Sanders, were charged with aiding and abetting a robbery affecting interstate commerce and the use or carriage of a firearm. At trial, the FBI...
-
The financial statements of P&G are presented in Appendix 5B or can be accessed at the books companion website, www.wiley.com/college/kieso. Instructions Refer to P&Gs financial statements and the...
-
1. [10] Is Grtzsch graph M(C5) Hamiltonian? Is the complement of M(C5) Hamilto- nian? Justify your answer.
-
I farm on a small scale. Last year, I started off by planting soybeans and corn. This year, the price of soybeans increased, and the price of corn remained the same. Naturally, I planted more corn...
-
Please make the response detailed and easy to read, thank you 1) During 2017, Smithfield Company introduced a new product carrying a two-year warranty against defects, which is included in the...
-
The table below shows recent worldwide market shares of producers of inkjet? printers: Firm Share of Worldwide Market Sales Brother 44?% Canon 1616 Dell 66 Epson 1818 ?Hewlett-Packard 4141 Lexmark...
-
How do individuals gain social capital through networks (see Gaining Power Through Social Networks); how do these translate into power and influence? Explain the dependence model. Describe the five...
-
How do you believe Tony Hsieh's life experiences and employment history influence his philosophies surrounding work environments? How would you describe the culture first created at Zappos? According...
-
A professor is expected to cover 16 chapters in an operations management text each semester. The class is scheduled to meet for one hour on Mondays, Wednesdays, and Fridays for 15 weeks. Because of...
-
What is demand forecasting? What are the importance and benefits of forecasting customer demand? Provide an overview of how a bank and a furniture manufacturer would forecast demand? In your response...
-
Determine the rotation at both supports (A and B) and deflection at free end (C) in the overhanging beam shown in Fig. 7. Assume the uniformly distributed load (w) to be equal to last two digits of...
-
Flicker, Inc., a closely held corporation, acquired a passive activity this year. Gross income from operations of the activity was $160,000. Operating expenses, not including depreciation, were...
-
Consider the following Lewis structure, where E is an unknown element: What are some possible identities for element E? Predict the molecular structure (including bond angles) for this ion. (See...
-
Calculate the internal energy change for each of the following. a. One hundred (100.) joules of work are required to compress a gas. At the same time, the gas releases 23 J of heat. b. A piston is...
-
Polystyrene can be made more rigid by copolymerizing styrene with divinylbenzene, What purpose does the divinylbenzene serve? Why is the copolymer more rigid? CH=CH2 CH=CH,
-
A phenotypically abnormal individual has a phenotypically normal father with an inversion on one copy of chromosome 7 and a phenotypically normal mother without any changes in chromosome structure....
-
Explain how polytene chromosomes of Drosophila are produced, and describe the six-armed structure they form.
-
Male honeybees, which are haploid, produce sperm by meiosis. Explain what unusual event (compared with other animals) must occur during spermatogenesis in honeybees to produce sperm. Does this...

Study smarter with the SolutionInn App


