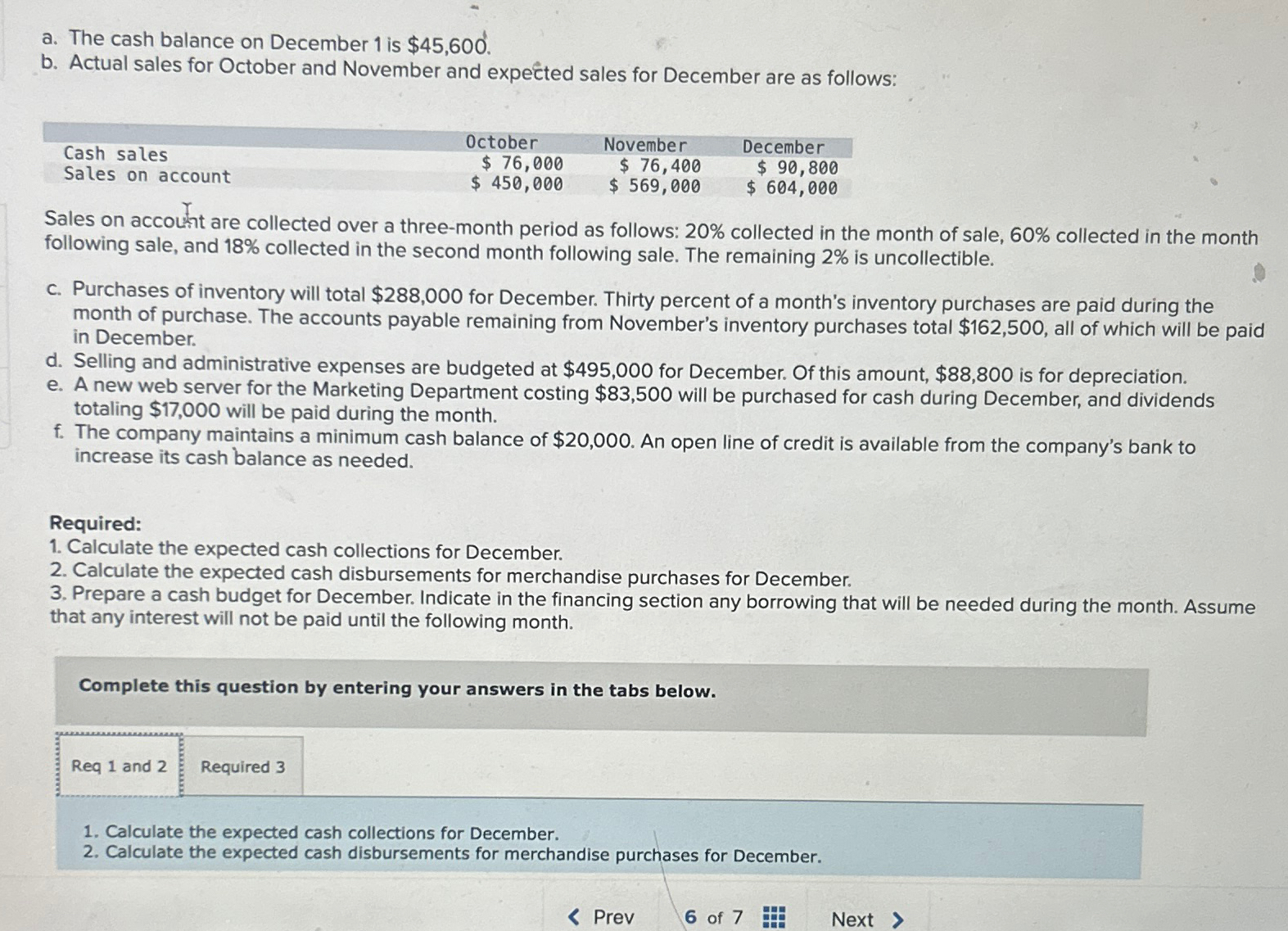
a. The cash balance on December 1 is $45,600. b. Actual sales for October and November...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
a. The cash balance on December 1 is $45,600. b. Actual sales for October and November and expected sales for December are as follows: Cash sales Sales on account October $ 76,000 $ 450,000 November $ 76,400 $ 569,000 December $ 90,800 $ 604,000 Sales on account are collected over a three-month period as follows: 20% collected in the month of sale, 60% collected in the month following sale, and 18% collected in the second month following sale. The remaining 2% is uncollectible. c. Purchases of inventory will total $288,000 for December. Thirty percent of a month's inventory purchases are paid during the month of purchase. The accounts payable remaining from November's inventory purchases total $162,500, all of which will be paid in December. d. Selling and administrative expenses are budgeted at $495,000 for December. Of this amount, $88,800 is for depreciation. e. A new web server for the Marketing Department costing $83,500 will be purchased for cash during December, and dividends totaling $17,000 will be paid during the month. f. The company maintains a minimum cash balance of $20,000. An open line of credit is available from the company's bank to increase its cash balance as needed. Required: 1. Calculate the expected cash collections for December. 2. Calculate the expected cash disbursements for merchandise purchases for December. 3. Prepare a cash budget for December. Indicate in the financing section any borrowing that will be needed during the month. Assume that any interest will not be paid until the following month. Complete this question by entering your answers in the tabs below. Req 1 and 2 Required 3 1. Calculate the expected cash collections for December. 2. Calculate the expected cash disbursements for merchandise purchases for December. < Prev 6 of 7 Next > a. The cash balance on December 1 is $45,600. b. Actual sales for October and November and expected sales for December are as follows: Cash sales Sales on account October $ 76,000 $ 450,000 November $ 76,400 $ 569,000 December $ 90,800 $ 604,000 Sales on account are collected over a three-month period as follows: 20% collected in the month of sale, 60% collected in the month following sale, and 18% collected in the second month following sale. The remaining 2% is uncollectible. c. Purchases of inventory will total $288,000 for December. Thirty percent of a month's inventory purchases are paid during the month of purchase. The accounts payable remaining from November's inventory purchases total $162,500, all of which will be paid in December. d. Selling and administrative expenses are budgeted at $495,000 for December. Of this amount, $88,800 is for depreciation. e. A new web server for the Marketing Department costing $83,500 will be purchased for cash during December, and dividends totaling $17,000 will be paid during the month. f. The company maintains a minimum cash balance of $20,000. An open line of credit is available from the company's bank to increase its cash balance as needed. Required: 1. Calculate the expected cash collections for December. 2. Calculate the expected cash disbursements for merchandise purchases for December. 3. Prepare a cash budget for December. Indicate in the financing section any borrowing that will be needed during the month. Assume that any interest will not be paid until the following month. Complete this question by entering your answers in the tabs below. Req 1 and 2 Required 3 1. Calculate the expected cash collections for December. 2. Calculate the expected cash disbursements for merchandise purchases for December. < Prev 6 of 7 Next >
Expert Answer:

Related Book For 

Managerial Accounting
ISBN: 9781259275814
11th Canadian Edition
Authors: Ray H Garrison, Alan Webb, Theresa Libby
Posted Date:
Students also viewed these accounting questions
-
Forecasts for next year for two firms, A and B are: Stock A Stock B Return on equity 17% 15% Earnings per share $ 5.00 $ 4.00 Dividends per share $ 3.00 $ 3.00 a. What are the dividend payout ratios...
-
Initial Population: Generation 1: Generation 2: Lab Genetic Algorithms Instructions Creating the Initial Population: The initial population was created using a random bit string generator. This step...
-
The hemispherical dome in Fig P2.91 weighs 30 kN and is filled with water and attached to the floor by six equally spaced bolts. What is the force in each bolt required to hold the dome down? cm e m...
-
Consider the three mutually exclusive projects that follow. The firm's MARR is 10% per year. a. Calculate each project's PW. b. Determine the IRR of each project. c. Which project would you...
-
In exchange for a $400 million fixed commitment line of credit, your firm has agreed to do the following: 1. Pay 1.9 percent per quarter on any funds actually borrowed. 2. Maintain a 4 percent...
-
How does the kinetic energy of the particle in Figure P24.59 change as it traverses the velocity selector? A. The kinetic energy increases. B. The kinetic energy does not change. C. The kinetic...
-
Firms J and K produce compact-disc players and compete against one another. Each firm can develop either an economy player (E) or a deluxe player (D). According to the best available market research,...
-
In this discussion you will on research ARM processor usage in desktop computers and servers, a trend that is gaining momentum. What immediate challenges does it present for Intel and AMD?
-
The following information has been taken from the books of Flirty Traders. Flirty Traders' financial year ends on 29 February and they are not a registered VAT Vendor. The following balances, amongst...
-
What is a Brnsted-Lowry acid? a proton acceptor a proton donor a hydroxide ion donor a hydroxide ion acceptor
-
5. A triglyceride has some polar and some non-polar bonds - and yet the molecule as a whole is very hydrophobic (i.e. water hating). a. Why is the tryiglyceride - as a whole - water hating? b....
-
Your research laboratory uses a sample of hydrochloric acid and obtains unexpected results. Your research advisor asks you to measure the concentration of this solution using a titration. Describe...
-
The Haber process is typically carried out at a temperature of approximately 500 C. What would happen to the rate of the forward reaction if the temperature were lowered to 100 C? View Available...
-
Suppose that one day all 330 million people in the US climb up on tables. At time t=0, we all jump off. The resulting shock, as we hit the Earth's surface, will start the entire Earth vibrating in...
-
Respond to the following: Think about a time in your life when you might have engaged in somewhat inappropriate behavior. Or, think about a time when you might have been at a crossroads in your life,...
-
Vince, Inc. has developed and patented a new laser disc reading device that will be marketed internationally. Which of the following factors should Vince consider in pricing the device? I. Quality of...
-
Let \(X, Y, X_{n}, Y_{n}: \Omega ightarrow \mathbb{R}, n \geqslant 1\), be random variables. a) If, for all n > 1, Xn Yn and if (Xn, Yn) (X, Y), then XIL Y. b) Let X Y such that X, Y ~ B1/2 = (80...
-
Let \(X_{n}, Y_{n}: \Omega ightarrow \mathbb{R}^{d}, n \geqslant 1\), be two sequences of random variables such that \(X_{n} \xrightarrow{d} X\) and \(X_{n}-Y_{n} \xrightarrow{\mathbb{P}} 0\). Then...
-
Let \(X_{n}, X, Y: \Omega ightarrow \mathbb{R}, n \geqslant 1\), be random variables. If \[\lim _{n ightarrow \infty} \mathbb{E}\left(f\left(X_{n}ight) g(Y)ight)=\mathbb{E}(f(X) g(Y)) \quad \text {...

Study smarter with the SolutionInn App


