A hydrocarbon (containing only carbon and hydrogen) with an unknown formula is subjected to combustion analysis....
Fantastic news! We've Found the answer you've been seeking!
Question:
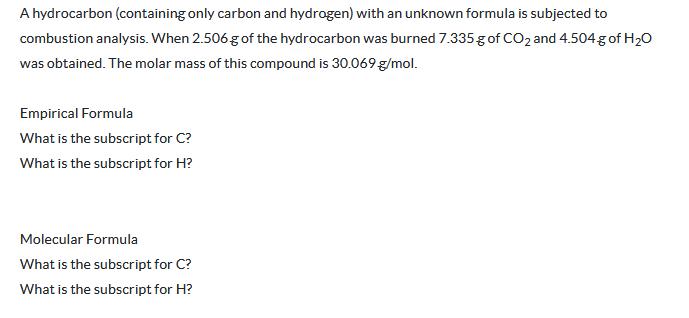
Transcribed Image Text:
A hydrocarbon (containing only carbon and hydrogen) with an unknown formula is subjected to combustion analysis. When 2.506 g of the hydrocarbon was burned 7.335 g of CO2 and 4.504 g of H₂O was obtained. The molar mass of this compound is 30.069 g/mol. Empirical Formula What is the subscript for C? What is the subscript for H? Molecular Formula What is the subscript for C? What is the subscript for H? A hydrocarbon (containing only carbon and hydrogen) with an unknown formula is subjected to combustion analysis. When 2.506 g of the hydrocarbon was burned 7.335 g of CO2 and 4.504 g of H₂O was obtained. The molar mass of this compound is 30.069 g/mol. Empirical Formula What is the subscript for C? What is the subscript for H? Molecular Formula What is the subscript for C? What is the subscript for H?
Expert Answer:
Answer rating: 100% (QA)
To find the empirical formula of the hydrocarbon we first need to determine the moles of carbon and ... View the full answer

Related Book For 

Vector Mechanics for Engineers Statics and Dynamics
ISBN: 978-0073212227
8th Edition
Authors: Ferdinand Beer, E. Russell Johnston, Jr., Elliot Eisenberg, William Clausen, David Mazurek, Phillip Cornwell
Posted Date:
Students also viewed these mechanical engineering questions
-
Draw temperature curves dry air parcels that we release with temperatures of 22C and 24C at the ground. The y-curve in the graph describes the environmental temperature profile. At what altitudes do...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The ABC Company has a cost of equity of 24.76 percent, a before-tax cost of debt of 5.37 percent, and a tax rate of 26 percent. What is the firm's weighted average cost of capital if the proportion...
-
Exercise 2-15 continues with the consulting business of Alex Kerr, begun in Serial Exercise 1-16. Here you will account for Kerr Consulting's transactions as it is actually done in practice. Kerr...
-
Use the master method to show that the solution to the binary-search recurrence T (n) = T (n/2) + (1) is T (n) = (lg n). Exercise 2.3-5 Referring back to the searching problem (see Exercise 2.1-3),...
-
This case arose after Felix DeWeldon, a well-known sculptor and art collector, sold three paintings to Robert McKean in 1994. Felix DeWeldon declared bankruptcy in 1991. In 1992, DeWeldon, Ltd.,...
-
(Accounting for Restricted Stock) Derrick Company issues 4,000 shares of restricted stock to its CFO, Dane Yapping, on January 1, 2010. The stock has a fair value of $120,000 on this date. The...
-
Listed below are eight firms that are currently operating in the market. The market share of each firm is listed in the table below. Firm Market Share Bird 16 Cat 11 Dog 8 Horse 4 Pig 5 Cow 10 Tiger...
-
David R. and Ella M. Cole (ages 39 and 38, respectively) are husband and wife who live at 1820 Elk Avenue, Denver, CO 80202. David is a self-employed consultant, specializing in retail management and...
-
Discuss the role of the judicial review in relation to administrative agencies.
-
Outline the specific functions provided by MYOB software. Discuss the problems of using a manual accounting system. Outline the benefits of using an online accounting software tool. Discuss three...
-
1.What was the premise behind the Recording Industry Association of Americas lawsuit against Napster? 2.Based on your knowledge of how Napster works, if you downloaded a copy of a song from a friends...
-
11) Short-term liquidity refers to an organization's ability to generate enough cash to repay long-term debts as they mature. 12) Long-term solvency refers to an organization's ability to meet...
-
Discuss the principles of pharmacogenomics and personalized medicine, highlighting how genetic variation influences drug metabolism, efficacy, and adverse reactions, and its implications for...
-
Ingrid Lederhaas-Okun was arrested on July 2, 2013, at her home in Connecticut on charges of wire fraud and interstate transportation of stolen property. She is accused of stealing jewelry from...
-
A 680-room business hotel has a potential room rate of $1,650. The hotel usually offers $1350 to FITs and $800 to groups. The fixed cost of each occupied room is $200. There is a 40-room group...
-
What impact has the Internet had on the globalization of small firms? How do you think small companies will use the Internet for business in the future?
-
A 2-kg sphere moving to the right with a velocity of 5 m/s strikes at A the surface of a 9-kg quarter cylinder which is initially at rest and in contact with a spring of constant 20 kN/m. The spring...
-
A 1.2-lb ball A is moving with a velocity vA when it is struck by a 2.4-lb ball B which has a velocity vB of magnitude vB = 18 ft/s. Knowing that the velocity of ball B is zero after impact and that...
-
For the beam and loading shown, determine (a) The distance a for which the maximum absolute value of the bending moment in the beam is as small as possible, (b) The corresponding value of M max. 14...
-
Using the data in Tables 7.13 and 7.14, does this farm qualify for debt (i.e., is the farm profitable, liquid, and solvent)? Table 7.13 Table 7.14 Item 2006 2007 2008 2009 2010 2011 2012 Gross cash...
-
The book basis of depreciable assets for Erwin Co. is 900,000 and the tax basis is 700,000 at the end of 2015. The enacted tax rate is 34% for all periods. Determine the amount of deferred taxes to...
-
How does an asset gain or loss develop in pension accounting?

Study smarter with the SolutionInn App


