(a) What is the % composition of the vapor in equilibrium with a boiling liquid that has...
Fantastic news! We've Found the answer you've been seeking!
Question:
(a) What is the % composition of the vapor in equilibrium with a boiling liquid that has a composition of 30% benzene and 70% toluene.
b). If a sample of vapor is found to have a composition of equal parts benzene and toluene, what is the composition of the boiling liquid that produced this vapor?

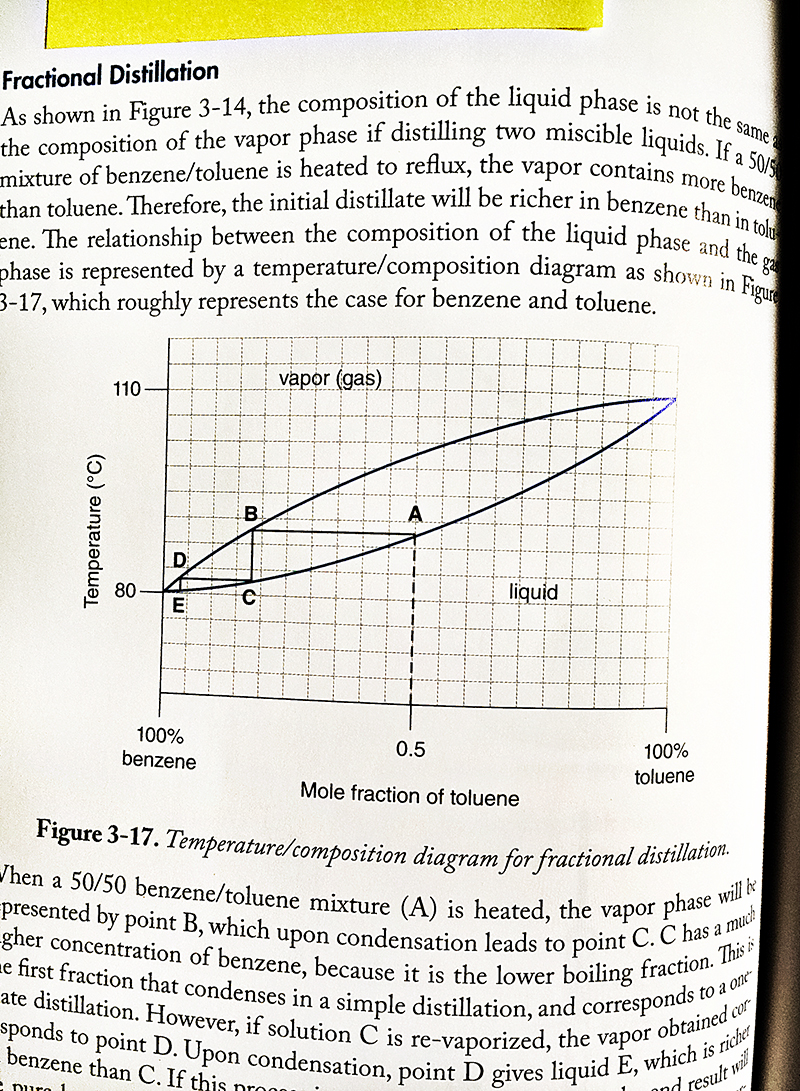
Transcribed Image Text:
Post-Lab Questions 1. Refer to MtC Figure 3-17 to answer the following questions. Note that each vertical line in the graph grid equals 5%, and the scale runs both ways for benzene and toluene: 100% benzene → 0% benzene 0% toluene → 100% toluene a. What is the % composition of the vapor in equilibrium with a boiling liquid that has a composition of 30% benzene and 70% toluene? b. If a sample of vapor is found to have a composition of equal parts benzene and toluene, what is the composition of the boiling liquid that produced this vapor? Fractional Distillation As shown in Figure 3-14, the composition of the liquid phase is not the same a the composition of the vapor phase if distilling two miscible liquids. If a 50/5 than toluene. Therefore, the initial distillate will be richer in benzene than in tolu mixture of benzene/toluene is heated to reflux, the vapor contains more benzene ene. The relationship between the composition of the liquid phase and the ga phase is represented by a temperature/composition diagram as shown in Figure 3-17, which roughly represents the case for benzene and toluene. Temperature (°C) 110 D E 100% benzene B vapor (gas) A 0.5 liquid Mole fraction of toluene 100% toluene Figure 3-17. Temperature/composition diagram for fractional distillation. When a 50/50 benzene/toluene mixture (A) is heated, the vapor phase will b gher concentration of benzene, because it is the lower boiling fraction. This is presented by point B, which upon condensation leads to point C. C has a much e first fraction that condenses in a simple distillation, and corresponds t ate distillation. However, if solution C is re-vaporized, the vapor obtained cor sponds to point D. Upon condensation, point D gives liquid E, which is richer to a one benzene than C. If this pron e Duro 1 and result will Post-Lab Questions 1. Refer to MtC Figure 3-17 to answer the following questions. Note that each vertical line in the graph grid equals 5%, and the scale runs both ways for benzene and toluene: 100% benzene → 0% benzene 0% toluene → 100% toluene a. What is the % composition of the vapor in equilibrium with a boiling liquid that has a composition of 30% benzene and 70% toluene? b. If a sample of vapor is found to have a composition of equal parts benzene and toluene, what is the composition of the boiling liquid that produced this vapor? Fractional Distillation As shown in Figure 3-14, the composition of the liquid phase is not the same a the composition of the vapor phase if distilling two miscible liquids. If a 50/5 than toluene. Therefore, the initial distillate will be richer in benzene than in tolu mixture of benzene/toluene is heated to reflux, the vapor contains more benzene ene. The relationship between the composition of the liquid phase and the ga phase is represented by a temperature/composition diagram as shown in Figure 3-17, which roughly represents the case for benzene and toluene. Temperature (°C) 110 D E 100% benzene B vapor (gas) A 0.5 liquid Mole fraction of toluene 100% toluene Figure 3-17. Temperature/composition diagram for fractional distillation. When a 50/50 benzene/toluene mixture (A) is heated, the vapor phase will b gher concentration of benzene, because it is the lower boiling fraction. This is presented by point B, which upon condensation leads to point C. C has a much e first fraction that condenses in a simple distillation, and corresponds t ate distillation. However, if solution C is re-vaporized, the vapor obtained cor sponds to point D. Upon condensation, point D gives liquid E, which is richer to a one benzene than C. If this pron e Duro 1 and result will
Expert Answer:
Answer rating: 100% (QA)
Solution a 65 Benzene and 35 Toluene Go up wards from the point of 30 Benz... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A freshly isolated sample of 90Y was found to have an activity of 9.8 105 disintegrations per minute at 1:00 p.m. on December 3, 2003. At 2:15 p.m. on December 17, 2003, its activity was...
-
What is the composition of the FASB's membership?
-
Is the composition of state legislatures in the House of Representatives related to the specific state? Use = 0.05. Perform the following steps. a. State the hypotheses and identify the claim. b....
-
solve the following instance of the 0/1 kropsack problem using dynamic programming approach. Assume that the knapsack capacity is 9. Item 1 3 4 weight 6 2 5 7 Benef $8 $4 $6 $10
-
The transactions that follow took place at the Lancaster Sports Arena during September 2013. This firm has indoor courts where customers can play tennis for a fee. It also rents equipment and offers...
-
A new public works project requires 300,000 hours of labor to complete. a. Suppose the labor market is perfectly competitive and the market wage is $20. What is the opportunity cost of the labor...
-
What would happen to the SML graph in Figure 8.8 if expected inflation increased or decreased? Figure 8.8 268 269 270 271 272 273 274 275 A Required Rate of Return TH-13.0% SML: r, RF+RPM * b D E F H...
-
Laminar flow in a triangular duct (Figure 3B.2) 2 one type of compact heat exchanger is shown in Figure 3B.2 (a). In order to analyze the performance of such an apparatus, it is necessary to...
-
How would you import a relational model design and generate a logical model from a relational model?
-
Allison, age 40, earns $81,915 annually; her wage replacement ratio has been determined to be 73%. She expects inflation will average 3% over her entire life expectancy. She expects to work until 67...
-
Q1 Assume the Fermi energy level is 0.03eV below the conduction band energy. (a) Determine the probability of state being occupied by an electron at E..(b) Repeat part (a) for an energy state at...
-
What would Ellen most likely recomended to the executive team with regard to trade-offs?
-
Quarko Co manufactures two products, Xerxes and Yoraths. Noinventories are held. The following data relates to the budget for eachunit of product. Fixed costs are $13,000 per quarter. Direct...
-
What might other firms learn from Walmart's experience regarding application forms, selection decisions, accommodations, management training, and other human resource management issues discussed in...
-
QUESTION: REQUIRED Use the information provided below to prepare the Statement of Comprehensive Income for the year ended 2 8 February 2 0 2 3 . INFORMATION FIESTA TRADERS PRE - ADJUSTMENT TRIAL...
-
What would the 4'P for a dental office be that provides Dental Crown, Teeth Whitening, Root Canal Dental, Hygiene Services, Dental Implant, Abscess Incision and Drainage, Cosmetic Procedures, Teeth...
-
Amelia has taken a loan to invest in a non-registered Canadian mutual fund portfolio and paid $2,000 in interest on that loan this year. She has also taken a loan to invest in her Registered...
-
Answer the following two independent questions. a. MM Corporation is considering several proposed investments for the coming budget year. MM produces electrical apparatus for industrial complexes....
-
Calculate the molality of each of the following solutions: (a) 14.3 g of sucrose (C12H22O11) in 676 g of water, (b) 7.20 moles of ethylene glycol (C2H6O2) in 3546 g of water.
-
A 250-mL glass bottle was filled with 242 mL of water at 20C and tightly capped. It was then left outdoors overnight, where the average temperature was - 5C. Predict what would happen. The density of...
-
A solution of 1.00 g of anhydrous aluminum chloride, AlCl3, in 50.0 g of water freezes at 21.11C. Does the molar mass determined from this freezing point agree with that calculated from the formula?...
-
Calculate the P(Gain) of each asset class (see above) and compare each. Which is most efficient?
-
Calculate and graphically demonstrate the risk of each asset; see above.
-
Compare the average returns for equity, debt and the major real estate asset classes for the last 5-, 10-, and 15-year periods. The asset classes are equity (S&P 500), debt (VBMFX), office, retail,...

Study smarter with the SolutionInn App


