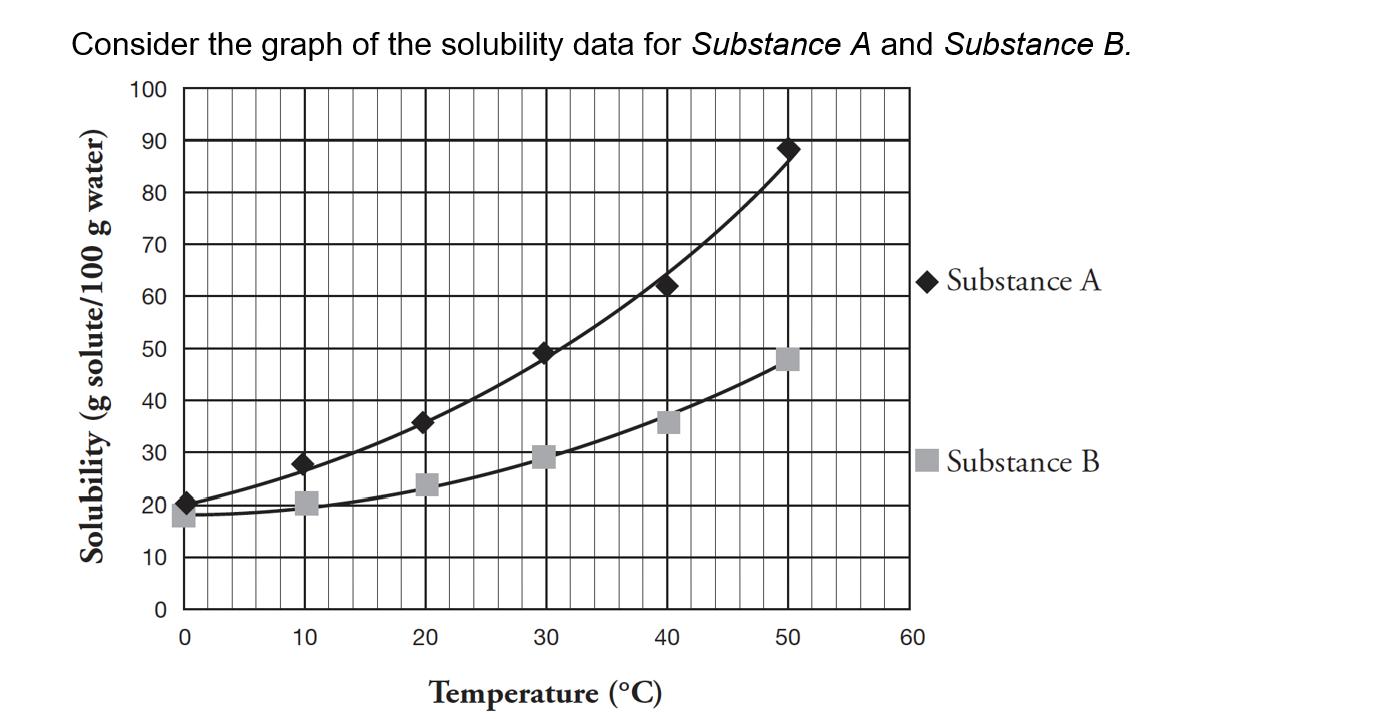
a. What is the solubility of Substance A in water at 20C in units of g/ml?...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
a. What is the solubility of Substance A in water at 20°C in units of g/ml? (2 points) b. If 35.0 g of Substance B is added to 100 ml of water at 30°C, what type of solution would result; unsaturated, saturated, or supersaturated? (2 points) Consider the graph of the solubility data for Substance A and Substance B. 100 Solubility (g solute/100 g water) 90 80 70 60 50 40 30 20 10 0 10 20 30 40 Temperature (°C) 50 60 Substance A Substance B a. What is the solubility of Substance A in water at 20°C in units of g/ml? (2 points) b. If 35.0 g of Substance B is added to 100 ml of water at 30°C, what type of solution would result; unsaturated, saturated, or supersaturated? (2 points) Consider the graph of the solubility data for Substance A and Substance B. 100 Solubility (g solute/100 g water) 90 80 70 60 50 40 30 20 10 0 10 20 30 40 Temperature (°C) 50 60 Substance A Substance B
Expert Answer:
Answer rating: 100% (QA)
Step 12 a the solubility of substance A in water at 20 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
The solubility of carbon dioxide in water is 0.161 g CO2 in 100 mL of water at 20C and 1.00 atm. A soft drink is carbonated with carbon dioxide gas at 5.50 atm pressure. What is the solubility of...
-
What is the solubility of magnesium hydroxide in a solution buffered at pH 8.80?
-
A 3.20-g sample of a salt dissolves in 9.10 g of water to give a saturated solution at 25C. What is the solubility (in g salt/100 g of H2O) of the salt?
-
A company has the following results for the three years to 31 October 2020: Assuming that all possible claims are made to relieve the trading loss against total profits, calculate the company's...
-
Suppose that we draw cards repeatedly and with replacement from a file of 100 cards, 50 of which refer to male and 50 to female persons. What is the probability of obtaining the second "female" card...
-
An axial-flow fan operates in sea-level air at \(1350 \mathrm{rpm}\) and has a blade tip diameter of \(3 \mathrm{ft}\) and a root diameter of \(2.5 \mathrm{ft}\). The inlet angles are...
-
How can computers be used to assist in the litigation process?
-
Capacity costs Kens Cornerspot, a popular university eatery in a competitive market, has seating and staff capacity to serve about 600 lunch customers every day. For the past two months, demand has...
-
Assume that the cost formula for one of a company's mixed expenses is $10,000+ $4.00 per unit. The company's planned level of activity was 2,000 units and its actual level of activity was 2,200...
-
Underground Sandwiches, a sandwich shop, has the following marginal physical product curve (labeled MPP) for its hourly production. (?) 20 18 16 O MPP and AP (Sandwiches per hour) AP MPP 0 1 3 5...
-
You are assigned the task of deciding how many tellers to employ for the 11:30-12:00 slot on a normal day. Assume that the cost of using a teller for an hour is $40. It is clear that customers are...
-
A producer of extruded plastic products finds that his mean daily inventory is 1,250 pieces. A new marketing policy has been put into effect and it is desired to test the null hypothesis that the...
-
Several square inches of gold leaf are required in the manufacture of a high-end component. Suppose that, the population of the amount of gold leaf has \(\sigma=8.4\) square inches. We want to test...
-
It is desired to test the null hypothesis \(\mu=30\) minutes against the alternative hypothesis \(\mu <30\) minutes on the basis of the time taken by a newly developed oven for \(n=50\) cakes baked....
-
In 64 randomly selected hours of production, the mean and the standard deviation of the number of acceptable pieces produced by a automatic stamping machine are \(\bar{x}=1,038\) and \(s=146\). At...
-
Refer to Exercise 2.34, page 46, concerning the number of board failures for \(n=32\) integrated circuits. A computer calculation gives \(\bar{x}=7.6563\) and \(s=\) 5.2216. At the 0.01 level of...
-
Compare and contrast the KMP and Rabin-Karp algorithms. Illustrate how the Rabin-Karp algorithm can be used to check if the given pattern is present in the given text. Given text: environmeironten...
-
Evaluate the integral, if it exists. Jo y(y + 1) dy
-
Thorium-230, which occurs in uranium minerals, decays by alpha emission to radium. Write the nuclear equation for this decay process.
-
Which salt would you expect to dissolve more readily in acidic solution, calcium phosphate, Ca3(PO4)2, or calcium sulfate, CaSO4? Explain.
-
A 0.400-g sample of propionic acid was dissolved in water to give 50.0 mL of solution. This solution was titrated with 0.150 M NaOH. What was the pH of the solution when the equivalence point was...
-
The output response of a first order system with excitation is: (a) \(y=x\left(1-e^{-t / \tau} ight)\) (b) \(y=x e^{-t / \tau}\) (c) \(y=x\left(1+e^{-t / \tau} ight)\) (d) \(y=x\left(1-e^{t \tau}...
-
What is an actuator? List various types of actuators.
-
What is a regulator?

Study smarter with the SolutionInn App


