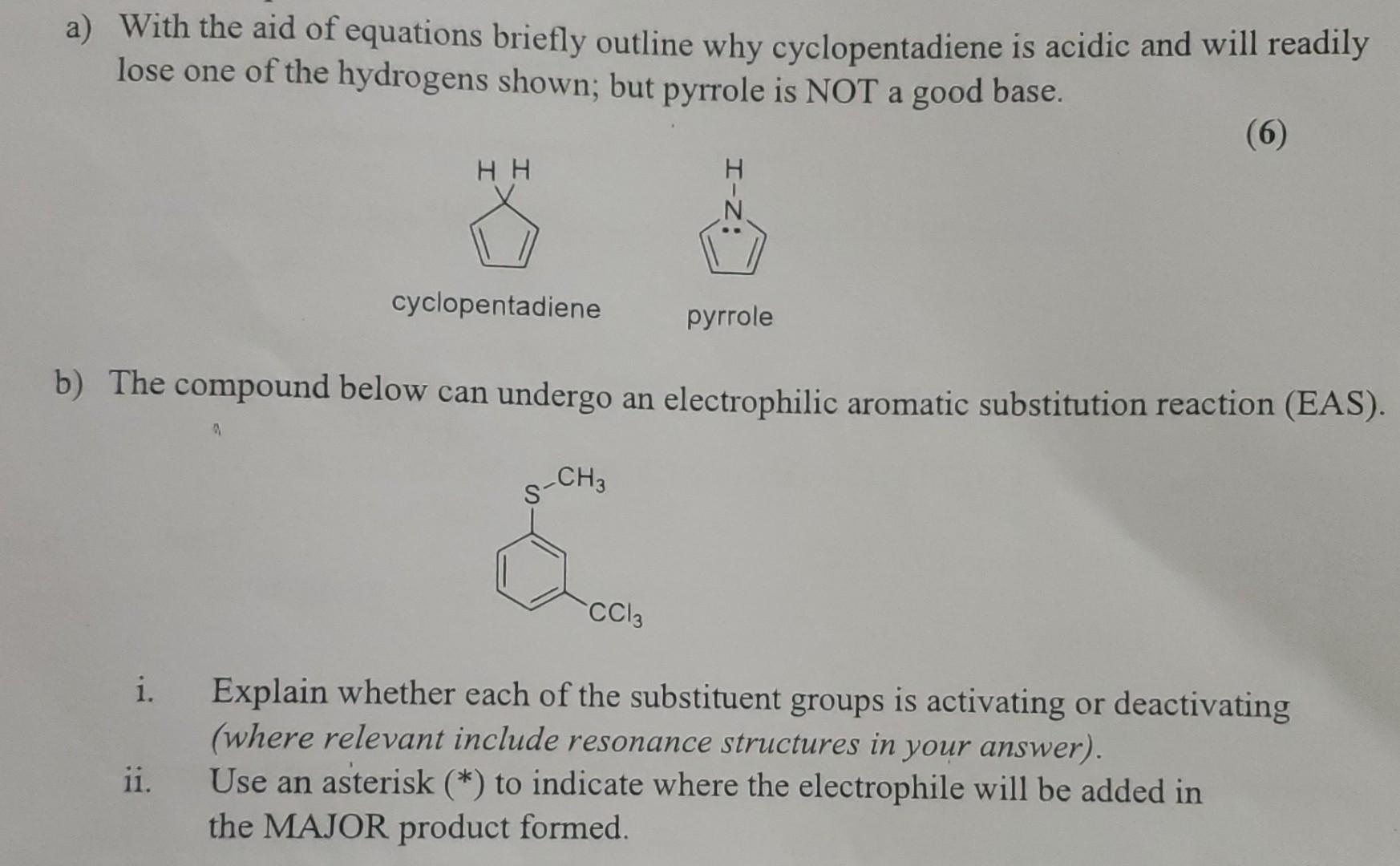
a) With the aid of equations briefly outline why cyclopentadiene is acidic and will readily lose...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
a) With the aid of equations briefly outline why cyclopentadiene is acidic and will readily lose one of the hydrogens shown; but pyrrole is NOT a good base. (6) i. ii. HH b) The compound below can undergo an electrophilic aromatic substitution reaction (EAS). a cyclopentadiene CH3 pyrrole CC13 Explain whether each of the substituent groups is activating or deactivating (where relevant include resonance structures in your answer). Use an asterisk (*) to indicate where the electrophile will be added in the MAJOR product formed. a) With the aid of equations briefly outline why cyclopentadiene is acidic and will readily lose one of the hydrogens shown; but pyrrole is NOT a good base. (6) i. ii. HH b) The compound below can undergo an electrophilic aromatic substitution reaction (EAS). a cyclopentadiene CH3 pyrrole CC13 Explain whether each of the substituent groups is activating or deactivating (where relevant include resonance structures in your answer). Use an asterisk (*) to indicate where the electrophile will be added in the MAJOR product formed.
Expert Answer:
Answer rating: 100% (QA)
Based on the provided questions lets tackle them one at a time a Cyclopentadiene is acidic due to the ability of the resulting anion to be stabilized ... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemical engineering questions
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
Create a use case diagram that would illustrate the use cases for the following dentist office system: Whenever new patients are seen for the first time, they complete a patient information form that...
-
The Clarkson Company produces engine parts for car manufacturers. A new accountant intern at Clarkson has accidentally deleted the company's variance analysis calculations for the year ended December...
-
a. Dynamic binding means that objects are allowed to take the form of either the class they are an instance of, or any of its subclasses. b. In an inheritance structure with a parent class Animal and...
-
Red Sky Delivery performs last-mile delivery services for online retailers such as Amazon. Red Sky employs delivery drivers who deliver packages to individual residence and business locations from a...
-
The following form is used by Matsuto Manufacturing Co. to compute payroll taxes incurred during April: Required: 1. Using the above form, calculate the employers payroll taxes for April. Assume that...
-
What is a personality trait you have in common with your parents? Perhaps it is a reaction you have in certain situations or something you find yourself saying. Is it a good or bad thing you picked...
-
A dog training business began on December 1. The following transactions occurred during its first month. December 1 Receives $23,000 cash as an owner investment in exchange for common stock. December...
-
How would you conduct Observational Research if you wanted to open a coffee shop in a mall? Provide at least 5 examples.
-
Part (a) Find the total energy transferred to the photon to four significant figures. Assume the reactants are initially at rest. Use the following atomic mass values when determining the total...
-
Focus on the current makeup of the health care workforce, the challenges posed by shortages in certain professions, and how these shortages are being addressed. Most health care in the United States...
-
A 15-kg uniform ladder 3.50 m long is placed against a smooth vertical wall. The ladder's base rests on a rough horizontal floor 2 m from the wall, with a coefficient of static friction equal to...
-
A dwarf planet orbits the Sun at a radius of 7.30*10^14 m. It has a mass of 6.84*10^22 kg. What is the gravitational force between this body and the sun as it orbits? The sun has a mass of 1.99 x 10...
-
During the year, Vilarium (Pty) Ltd lost two laptops together with three hard drives that had back-up data due to theft. There were reports of two men having been seen suspiciously loitering around...
-
What is the role of primary market in fund transferring from providers to users?
-
Federated Shipping, a competing overnight delivery service, informs the customer in Problem 65 that they would ship the 5-pound package for $29.95 and the 20-pound package for $59.20. (A) If...
-
Olive Corporation was formed and began operations on January 1, 2012. The corporation's income statement for the year and the balance sheet at year-end are presented below. The corporation made...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Abigail (Abby) Boxer is a single mother working as a civilian accountant for the U.S. Army. Her Social Security number is 676-73-3311 and she lives at 3456 Alamo Way, San Antonio, TX 78249. Helen,...
-
While the BohrSommerfeld condition sometimes gets the energy eigenvalues exactly correct, it can also be used for systems where the exact solution is not known. In this example, we will estimate the...
-
It's useful to see how our quantum perturbation theory works in a case that we can solve exactly. Let's consider a two-state system in which the Hamiltonian is...
-
Let's see how the variational method works in another application. Let's assume we didn't know the ground-state energy of the quantum harmonic oscillator and use the variational method to determine...

Study smarter with the SolutionInn App


