Question: (a) Write a mathematical expression that can be used to determine the value of S, the molar solubility of Pbl2(s). (b) If Pbl2(s) is
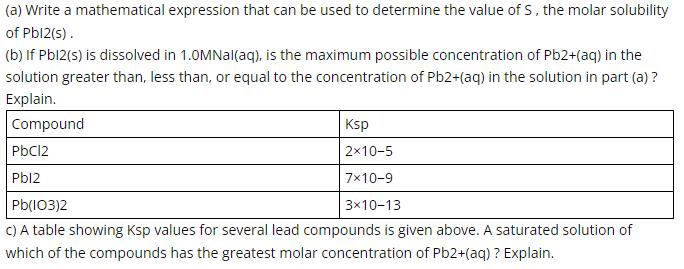
(a) Write a mathematical expression that can be used to determine the value of S, the molar solubility of Pbl2(s). (b) If Pbl2(s) is dissolved in 1.0MNal(aq), is the maximum possible concentration of Pb2+(aq) in the solution greater than, less than, or equal to the concentration of Pb2+(aq) in the solution in part (a) ? Explain. Compound Ksp PbC12 2x10-5 Pbl2 7x10-9 3x10-13 C) A table showing Ksp values for several lead compounds is given above. A saturated solution of Pb(I03)2 which of the compounds has the greatest molar concentration of Pb2+(aq) ? Explain.
Step by Step Solution
3.38 Rating (151 Votes )
There are 3 Steps involved in it
Answer The mixing of the twocomponent is called solutions The two ... View full answer

Get step-by-step solutions from verified subject matter experts


