Question: Above is a concentration cell for measuring Ksp for silver bromide. Given that the voltage for this cell at 298K is E=0.739 V, and

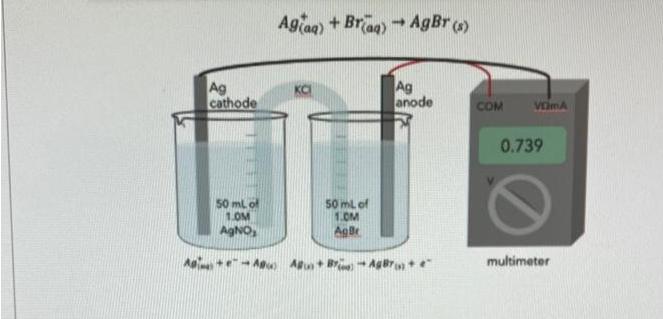
Above is a concentration cell for measuring Ksp for silver bromide. Given that the voltage for this cell at 298K is E=0.739 V, and that the overall reaction in this cell concentration cell is Use the data above to calculate pKsp, at equilibrium where pKsp = -log(Ksp) (note that when set-up with a voltmeter like this no significant current is drawn and the system reaches equilibrium) Ag cathode 50 mL of 1.0M AgNO, Agiaq) + Braq) = AgBr (s) + Ag anode 50 mL of 1.0M AgBr A+A) A+B-AgBr+ e COM VEMA 0.739 multimeter
Step by Step Solution
3.41 Rating (145 Votes )
There are 3 Steps involved in it
To calculate pKs we must first identify the reaction that occurs in the concentration cell The volta... View full answer

Get step-by-step solutions from verified subject matter experts


