The compound cyclopentanone has the molecular formula C,H,O. The C-NMR spectrum has three distinct peaks, at...
Fantastic news! We've Found the answer you've been seeking!
Question:
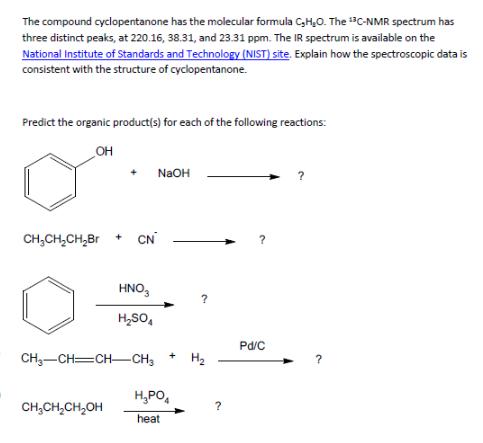
Transcribed Image Text:
The compound cyclopentanone has the molecular formula C,H,O. The C-NMR spectrum has three distinct peaks, at 220.16, 38.31, and 23.31 ppm. The IR spectrum is available on the National Institute of Standards and Technology (NIST) site. Explain how the spectroscopic data is consistent with the structure of cyclopentanone. Predict the organic product(s) for each of the following reactions: OH + NaOH CH3CH2CH2Br + CN HNO3 H₂SO4 CH—CH=CHCH3 Pd/C + H₂ H₁PO CH₂CH₂CH₂OH ? heat ? The compound cyclopentanone has the molecular formula C,H,O. The C-NMR spectrum has three distinct peaks, at 220.16, 38.31, and 23.31 ppm. The IR spectrum is available on the National Institute of Standards and Technology (NIST) site. Explain how the spectroscopic data is consistent with the structure of cyclopentanone. Predict the organic product(s) for each of the following reactions: OH + NaOH CH3CH2CH2Br + CN HNO3 H₂SO4 CH—CH=CHCH3 Pd/C + H₂ H₁PO CH₂CH₂CH₂OH ? heat ?
Expert Answer:
Answer rating: 100% (QA)
To determine if the spectroscopic data is consistent with the structure of cyclopentanone CHO we can analyze the information provided 1 3CNMR Spectrum ... View the full answer

Related Book For 

Advanced Accounting
ISBN: 978-0077431808
10th edition
Authors: Joe Hoyle, Thomas Schaefer, Timothy Doupnik
Posted Date:
Students also viewed these finance questions
-
OTIFIHindi Exam Fall 2022 Excel Sign Insert Page Layout Formulas Data Review View Help Tell me what you want to do Calibri 12 BIU ' ' A A 4 A 4 ab Wrap Text AutoSum Number Fill 4 Merge & Center 4 $...
-
6. Emily walks every day, and she keeps a record of the number of miles she walks each day. The histogram and five-number summary below were created from the recorded miles for a random sample of 25...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Ice Cool produces two different models of air conditioners. The activities, costs, and cost drivers associated with the production processes follow. Budgeted Activity Usage 7,600 Activity Machining...
-
Find the shortest distance between the following pairs of nonparallel lines and find the points on the lines that are closest together. (a) [x y z]T = [1 -1 0]T + s[1 1 1]T [x y z]T = [2 -1 3]T + t[3...
-
Consider the below information: Two period, two state world. Let the current stock price be 90 The risk-free rate be 8%. Each period the stock price can go either up by 10% or down by 8%. A call...
-
Charles Smith, Inc., is a manufacturer of small office equipment. Smith transacts most of its business on credit and offers its customers credit terms of 2/10, n/30. On July 1, Smith shipped an order...
-
Jon Williams, CPA, is in the middle of the real- life soap opera, Taxing Days of Our Lives. The Cast of Characters Oneway Corporation is Williamss audit and tax client. The three directors are the...
-
The following is a partial performance report for a revenue center for the Southern Division of B sion of Flower City Restaurants. (Click the icon to view the financial results.) Fill in the missing...
-
As the manager of credit card services at Bank of Hanover (BOH), you're aware that the average profitability of a credit card customer grows with the number of years they have used the credit card....
-
1. Demonstrate your ability to solve equations with various function types 2. Demonstrate your ability to solve multivariable equations for a specified variable 3. Demonstrate your ability to solve...
-
Shaw Incorporated began this period with a budget for 1,040 units of predicted production. The budgeted overhead at this predicted activity follows. At period-end, total actual overhead was $96,400,...
-
Three point charges, q = -4.07nC, 92 = 4.97nC and 93 = 2.97nC are aligned along the x axis as shown in the figure below. Assume that L = 0.493m and L = 0.838m. * 4 L -X 9 92 Calculate the electric...
-
Balances on March 31 Direct materials used (in March) Direct labor used (in March) Overhead applied (March) Costs during April Direct materials used Direct labor used Overhead applied Status on April...
-
A student is doing a physics experiment with a pendulum in the car ride home from school. At one point during the ride she notices that the 1.7kg bob of the pendulum is holding the position shown....
-
Let S be a bounded infinite set, and let x = sup(s). Prove that if x S then x is an accumulation point of s.
-
What market efficiency suggests that at a minimum, the current price of the stock reflects the stock's own past prices?Explain
-
a. Determine the domain and range of the following functions.b. Graph each function using a graphing utility. Be sure to experiment with the window and orientation to give the best perspective of the...
-
Livingston Company is a wholly owned subsidiary of Rose Corporation. Livingston operates in a foreign country with financial statements recorded in goghs (GH), the companys functional currency....
-
Why were several original standards issued by the IASC revised in 1993?
-
A client, Beth Voga, asks for advice. She tells you that her grandmother, a widowed resident of Montana, has no will. She asks whether any portion of her grandmothers estate will pass to her (Beths)...
-
Claire Corporations trial balance includes the following expenses: Assuming no change in the work-in-process and finished goods inventory balances for the year, what total amount should Claire report...
-
Carson Company reported the following amounts for December: What amount of raw materials was used in production in December? a. $71,000 b. $73,000 c. $77,000 d. $85,000 Beginning raw materials...
-
Carson Company reported the following amounts for December: What is cost of goods manufactured for December? a. $176,000 b. $182,000 c. $186,000 d. $188,000 Beginning raw materials inventory....

Study smarter with the SolutionInn App


