Q3). Q4. A company shows the following financial indicators on its balance sheet: : Acid-test Ratio, 1;
Fantastic news! We've Found the answer you've been seeking!
Question:
Q3).
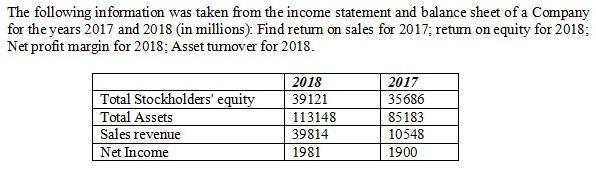
Q4.

Transcribed Image Text:
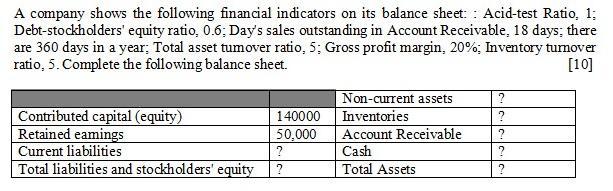
A company shows the following financial indicators on its balance sheet: : Acid-test Ratio, 1; Debt-stockholders' equity ratio, 0.6; Day's sales outstanding in Account Receivable, 18 days; there are 360 days in a year; Total asset tumover ratio, 5; Gross profit margin, 20%; Inventory turnover ratio, 5. Complete the following balance sheet. [10] Non-current assets ? Contributed capital (equity) Retained eamings Current liabilities Total liabilities and stockholders' equity ? 140000 Inventories Account Receivable Cash 50,000 ? Total Assets ? A company shows the following financial indicators on its balance sheet: : Acid-test Ratio, 1; Debt-stockholders' equity ratio, 0.6; Day's sales outstanding in Account Receivable, 18 days; there are 360 days in a year; Total asset tumover ratio, 5; Gross profit margin, 20%; Inventory turnover ratio, 5. Complete the following balance sheet. [10] Non-current assets ? Contributed capital (equity) Retained eamings Current liabilities Total liabilities and stockholders' equity ? 140000 Inventories Account Receivable Cash 50,000 ? Total Assets ?
Expert Answer:
Answer rating: 100% (QA)
Answer 1 Assets Amount Liabilities and Stockholder Equity Amount Non Current Assets 19000000 Equity 14000000 Inventories 21920000 Retained Earnings 50... View the full answer

Related Book For 

Financial Accounting Tools for business decision making
ISBN: 978-0470534779
6th Edition
Authors: Paul D. Kimmel, Jerry J. Weygandt, Donald E. Kieso
Posted Date:
Students also viewed these accounting questions
-
The following information was taken from the income statement of AJAX Corporation for the year ended December 31, 2014. AJAX is a holding company with subsidiaries providing research, development,...
-
The following data are taken from the income statement and balance sheet of Keaner Machinery, Inc.: Using this information, prepare a partial statement of cash flows for the year ended December 31,...
-
From the income statement and balance sheet of Alexander Company (Figures 22.16 and 22.17), compute the following for 2013: ALEXANDER COMPANY COMPARATIVE INCOME STATEMENT FOR YEARS ENDED DECEMBER 31,...
-
Write the boldfaced numbers in each problem in scientific notation. The largest of the 50 United States is Alaska, with land area of about 365,482,000 acres, while the smallest is Rhode Island, with...
-
What is specific performance?
-
Assume that we will use the sample data from Exercise 1 Video Games with a 0.05 significance level in a test of the claim that the population mean is greater than 90 sec. If we want to construct a...
-
The adjusted trial balance for Country Cookin Catering, Inc., is presented next. Prepare the income statement, statement of retained earnings, and balance sheet for Country Cookin Catering, Inc., for...
-
The comparative financial statements of Kenmore Pools. Inc. for 2017, 2016, and 2015 included the following select data: Requirements 1. Compute these ratios for 2017 and 2016: a. Current ratio b....
-
Suppose you observed returns of -5%, 12%, 5%, and 10% for a stock overthe past four years. What is the standard deviation of the stock'sreturns based on historical data?
-
Background: In the initial contract negotiation stage, the contract price with SM was $10.1 million in cash. However, as part of the final contract negotiations, SM agreed to give EYE SPY its old...
-
Use the normal distribution of SAT critical reading scores for which the mean is 502 and the standard deviation is 120. Assume the variable x is normally distributed. (a) What percent of the SAT...
-
Shiller Company received a bank statement showing the account balance $268,500, which is different from the amount shown in Shiller's general ledger. The following events account for the discrepancy...
-
At the beginning of this month, Friedman Company established a petty cash fund with the amount of \($10,000.\) At the end of this month, it was found that the balance of this fund was \($490,\) but...
-
Suppose Wal-Mart accepts from Apple Stores a \$15,000, 3-month, 9\% note dated September 30 in settlement of Apple's overdue account. (1) What is the maturity date of the note? (2) Assuming at the...
-
For a sample time of 0. 005 s, a. evaluate z at 10 Hz. b. Repeat for 20 Hz. c. Repeat for 50 Hz.
-
Does size matter? Warehouse clubs such as Costco, BJs, and Sams Club employ a strategy that includes large product sizes with low per unit costs. Suppose a supplier, such as Clorox, decides to sell...
-
Kaitline, a CPA, prepared Jared's tax return and was paid $2,000. Jared instructed Kaitline not to report some income he had received for helping a neighbor build a deck on his house. Kaitline...
-
Refer to the Conservation Ecology (Dec. 2003) study of the causes of forest fragmentation, presented in Exercise 2.166 (p. 97). Recall that the researchers used advanced high-resolution satellite...
-
Summary financial information for Newburg Company is as follows. Compute the amount and percentage changes in 2013 using horizontal analysis, assuming2012 is the base year. Dec. 31, 2012 Dec. 31,...
-
Mandall Company constructed a warehouse for $280,000. Mandall estimates that the warehouse has a useful life of 20 years and no residual value. Construction records indicate that $40,000 of the cost...
-
The February 13, 2010, issue of the Wall Street Journal includes an article by Scott Thurm entitled For Some Firms, a Case of Quadrophobia. Instructions Read the article and answer the following. (a)...
-
Calculate the acentric factor for ethanol. The vapour pressure of ethanol can be estimated from the following equation: \[ \log _{10} P^{\text {Sat }}=8.1122-\frac{1592.864}{t+226.184} \] where...
-
For liquid acetone at \(20^{\circ} \mathrm{C}\) and \(1 \mathrm{bar}\), \[ \beta=1.487 \times 10^{-3} /{ }^{\circ} \mathrm{C} \quad \alpha=62 \times 10^{-6} / \mathrm{bar} \quad V=1.287...
-
Determine the molar volume of ammonia vapour and ammonia liquid at \(321.55 \mathrm{~K}\) and 1.95 \(\mathrm{MPa}\). Ammonia is assumed to follow van der Waals equation of state.

Study smarter with the SolutionInn App


