To calculate your Average value of R: Make sure you've calculated number of moles of H...
Fantastic news! We've Found the answer you've been seeking!
Question:

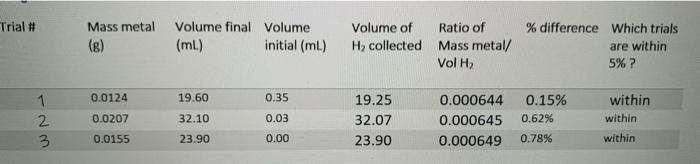
Transcribed Image Text:
To calculate your Average value of R: Make sure you've calculated number of moles of H₂ correctly Make sure you know Volumes of H₂ collected • Make sure you know the Pressure of H₂ (remember to account for water vapour pressure) • Make sure you know the Temperature of H₂ (the measured temperature of the room). Calculate the value of R for trials that were within 5% difference and Calculate your Average Value of R. . . Trial # 1 wN. 2 3 Mass metal (g) 0.0124 0.0207 0.0155 Volume final Volume (ml) 19.60 32.10 23.90 initial (ml) 0.35 0.03 0.00 Volume of H₂ collected 19.25 32.07 23.90 Ratio of Mass metal/ Vol H₂ 0.000644 0.000645 0.000649 % difference Which trials are within 5% ? 0.15% 0.62% 0.78% within within within To calculate your Average value of R: Make sure you've calculated number of moles of H₂ correctly Make sure you know Volumes of H₂ collected • Make sure you know the Pressure of H₂ (remember to account for water vapour pressure) • Make sure you know the Temperature of H₂ (the measured temperature of the room). Calculate the value of R for trials that were within 5% difference and Calculate your Average Value of R. . . Trial # 1 wN. 2 3 Mass metal (g) 0.0124 0.0207 0.0155 Volume final Volume (ml) 19.60 32.10 23.90 initial (ml) 0.35 0.03 0.00 Volume of H₂ collected 19.25 32.07 23.90 Ratio of Mass metal/ Vol H₂ 0.000644 0.000645 0.000649 % difference Which trials are within 5% ? 0.15% 0.62% 0.78% within within within
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Andy is further advised by the real estate agent that the estimated amount of stamp duty for the purchase of this flat is $198,000, the property insurance premium is $700 per month, the monthly...
-
Calculate the number of moles of KOH in 5.50 mL of a 0.360 M KOH solution. What is the pOH of the solution?
-
Calculate the volume in mL of a 1.420 M NaOH solution required to titrate the following solutions: (a) 25.00 mL of a 2.430 M HCl solution (b) 25.00 mL of a 4.500 M H2 SO4 solution (c) 25.00 mL of a...
-
What is the leading cause of death among people with HIV / AIDS in Africa?
-
Kevin is an employee of One Hour Dry Cleaners, Inc. All employees of One Hour are eligible for a 40 percent discount on their dry cleaning. During the year, Kevin paid $300 for cleaning that normally...
-
If H = yax xay A/m on plane z = 0, (a) Determine the current density and (b) Verify Ampere's law by taking the circulation of H around the edge of the rectangle Z = 0, 0 < x < 3, 1 < y < 4.
-
Suppose that the recycle times of two cameras are recorded. The extension of the positive real line \(R\) is to take the sample space to be the positive quadrant of the plane \[ S=R^{+} \times R^{+}...
-
Calculate the following ratios for Lake of Egypt Marina, Inc. as of year-end 2018? Use the following financial statements for Lake of Egypt Marina, Inc. Calculate the following ratios for Lake of...
-
1. What is tension? 2. What does tension do to materials? 3. What is compression? 4. What does compression do to materials?
-
Name the bones of the pectoral girdle and upper limb?
-
The pH of a 0.551 M solution of sodium methanoate, HCOO Na*, is 8.75. Calculate K, for the methanoate ion, HCOO". 1.0x105 5.7x1011 1.8x104 5.7x10 18 O 3.2x109
-
Compare the value derived from your computation with the price traded in the stock market around the time the 10-K report for the most recent period was filed. What levels of valuation parameters...
-
Forrest, the CMA of Acme Widgets Corp., sets the spending limit for his teams and requires all spending to stay within the promotional budget. What type of budgeting is being used by Widgets Corp?
-
There are a number of places where a motivated valuation could produce a higher final value. Let's take a look at some of the ways the shareholders' expert disagreed with the expert for Silver Lake...
-
How do second-wave cognitive-behavioural therapies differ from the traditional cognitive-behavioural therapies of Beck and Ellis?
-
Calculate the assessment ratio for all properties, which reflects the ratio of a parcel's assessed value to its market value. Use the parcel's most recent sale price as a proxy for its current market...
-
Assume that the holding cost of inventory at Macys is 35% per year. Assume further that Macys turns its inventory 3 times per year. How much does it cost Macys to hold a dress that it purchased for...
-
A circular concrete shaft liner with Youngs modulus of 3.4 million psi, Poissons ratio of 0.25, unconfined compressive strength 3,500 psi and tensile strength 350 psi is loaded to the verge of...
-
NaF has the same structure as NaCl. (a) Use ionic radii from Chapter 7 to estimate the length of the unit cell edge for NaF. (b) Use the unit cell size calculated in part (a) to estimate the density...
-
(a) Which of the thermodynamic quantities T, E, q, w, and S are state functions? (b) Which depend on the path taken from one state to another? (c) How many reversible paths are there between two...
-
Referring to Figure 11.29, describe the phase changes (and the temperatures at which they occur) when CO2 is heated from - 80 oC to - 20 oC at (a) A constant pressure of 3 atm, (b) A constant...
-
Give examples of policies that a country could implement to reduce its absorption.
-
In Section 6.2.3, we made the comment "Perception is reality." How does this play in the valuation of a tangible versus intangible asset? 6.2.3 Politics to explore Our roadmap for this chapter would...
-
We discussed risk aversion as being descriptive of investor behavior. Can Do you think of any real-world behavior that you might consider to be evidence of the existence of risk preferrers?

Study smarter with the SolutionInn App


