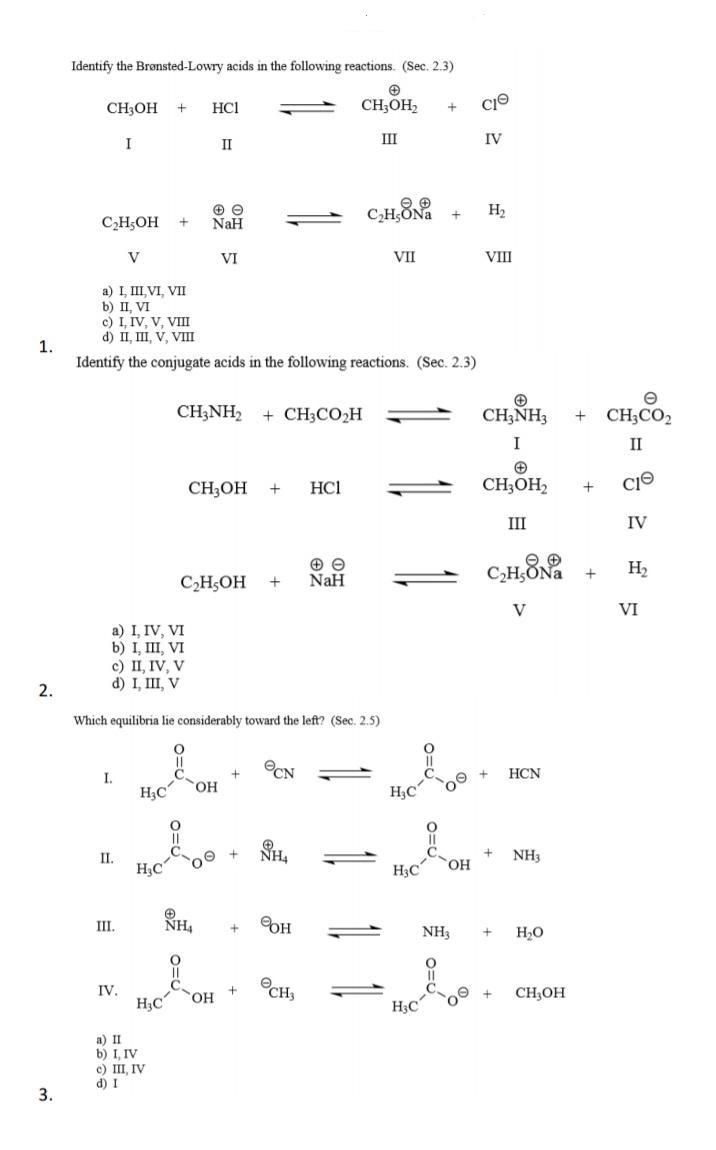
Identify the Brnsted-Lowry acids in the following reactions. (Sec. 2.3) CH3OH HCI CH;OH2 III IV II...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Identify the Brønsted-Lowry acids in the following reactions. (Sec. 2.3) CH3OH HCI CH;OH2 III IV II H2 CH;OH NaH V VI VII VII a) I III VI, VII b) II, VI c) I. IV. V. VII d) II, III, V, VIII 1. Identify the conjugate acids in the following reactions. (Sec. 2.3) CH;NH2 + CH3CO2H CH;NH3 CH,CO, I II CH;OH HCI CH;OH2 II IV C,HONa H2 CH;OH NaH V VI a) I, IV, VI b) I, III, VI c) II, IV, V d) I, III, V 2. Which equilibria lie considerably toward the left? (Sec. 2.5) OCN HCN I. H,C H,C NH, NH3 П. H3C oe + H3C HO, III. NH, OH NH3 H,O OCH, IV. HO, CH,OH H3C H;C a) II b) I, IV c) III, IV d) I 3. + + Identify the Brønsted-Lowry acids in the following reactions. (Sec. 2.3) CH3OH HCI CH;OH2 III IV II H2 CH;OH NaH V VI VII VII a) I III VI, VII b) II, VI c) I. IV. V. VII d) II, III, V, VIII 1. Identify the conjugate acids in the following reactions. (Sec. 2.3) CH;NH2 + CH3CO2H CH;NH3 CH,CO, I II CH;OH HCI CH;OH2 II IV C,HONa H2 CH;OH NaH V VI a) I, IV, VI b) I, III, VI c) II, IV, V d) I, III, V 2. Which equilibria lie considerably toward the left? (Sec. 2.5) OCN HCN I. H,C H,C NH, NH3 П. H3C oe + H3C HO, III. NH, OH NH3 H,O OCH, IV. HO, CH,OH H3C H;C a) II b) I, IV c) III, IV d) I 3. + +
Expert Answer:
Answer rating: 100% (QA)
1 option b is the answer Because Bronsted Lowry acid means proton H donor Hence ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
Which of the following reactions involve homogeneous equilibria and which involve heterogeneous equilibria? Explain the difference. a. 2NO(g) +02(8) 2NO2(8) C. 2N200g)2N2(g) + 02(g) d. 2NH(g)...
-
A small block slides at a speed d v = 3 m/s on a horizontal surface at a height h = 1 m above the ground. Determine (a) The angle at which it will leave the cylindrical surface BCD, (b) The distance...
-
1. Two football fans are listening to the Grey Cup game on the radio, one in Montreal, where the game is being played, the other in Vancouver, 3692 km away. How much sooner does the Montreal fan hear...
-
On January 15, Sheridan Service received a shipment with an invoice dated January 14, terms 4/10 E.O.M., for $2592. On February 9, Sheridan Service mailed a cheque for $1392 in partial payment of the...
-
What is Augmented and Virtual Reality?
-
The English scores for a sample of 14 students Find the mean, the median, and the mode of the data, if possible. If any measure cannot be found or does not represent the center of the data, explain...
-
Ashley owns a retail shoe store that is insured for $120,000 under the business income (and extra expense) coverage form. Because of a fire, Ashley was forced to close the store for three months....
-
Oriole Company has the following production data for selected months. Ending Work in Process Beginning Month Work in Process Units Transferred Out % Complete as to Units Conversion Cost January -0-...
-
An increasing number of Americans are moving to a warmer climate when they retire. To take advantage of this trend, Sunny Skies Unlimited is undertaking a major real estate development project. The...
-
Do you use social media to communicate? What are the strengths and weaknesses of Facebook, Twitter and Instagram? How can you use LinkedIn to advance your career?
-
Suspend a wire grill from a refrigerator or an oven from a string, holding the ends of the string to your ears. Let a friend gently stroke the grill with pieces of broom straw and with other objects....
-
The Korvette concept was started and run by one person and his group of friends. How could its failure have been avoided? Was the problem one of strategy (overexpansion), or was it organizational?...
-
Suppose \(\mathbf{x} \sim M N(n, \boldsymbol{\pi})\) follows a multinomial distribution of size \(n\) and probability \(\pi\). Derive the variance matrix of \(\mathbf{x}\).
-
Consider PowerBar, the strategy for which is summarized in the PowerBar case. What implications for the culture, structure, systems, and people would you suggest given the nature of the product and...
-
What performance measure would you consider most important for McDonald's? For Chevrolet?
-
Describe how relational database management systems are expanding their capabilitiesin the market place to address the growing needs of multi-model, NoSQL and NewSQL a) Define what multi-model is and...
-
The following data are supplied for the common stocks of Nikola Corporation, Tesla, Inc. and General Motors: Nikola Corp (NKLA) Tesla Inc. (TSLA) Close Price ($) Close Price ($) 67.53 30.00 40.81...
-
Molecular orbital calculations may be used to predict the dipole moments of molecules. (a) Using molecular modeling software and the computational method of your choice, calculate the dipole moment...
-
33S has a nuclear spin of% and a nuclear g-factor of 0.4289 calculate the energies of the nuclear spin states in a magnetic field of7.500 T.
-
The particle on a ring is a useful model for the motion of electrons around the porphine ring (2), the conjugated macro cycle that forms the structural basis of the haem group and the chlorophylls....
-
Find each probability using the standard normal distribution. (a) \(P(z>-1.68)\) (b) \(P(z <2.23)\) (c) \(P(-0.47
-
Find the probability that a randomly selected person has an IQ score between 95 and 105. Is this an unusual event? Explain. In a standardized IQ test, scores are normally distributed, with a mean...
-
The random variable \(x\) is normally distributed with the given parameters. Find each probability. (a) \(\mu=9.2, \sigma \approx 1.62, P(x <5.97)\) (b) \(\mu=87, \sigma \approx 19, P(x>40.5)\) (c)...

Study smarter with the SolutionInn App


