What is the stronger acid in the following reaction if the equilibrium constant is much less...
Fantastic news! We've Found the answer you've been seeking!
Question:
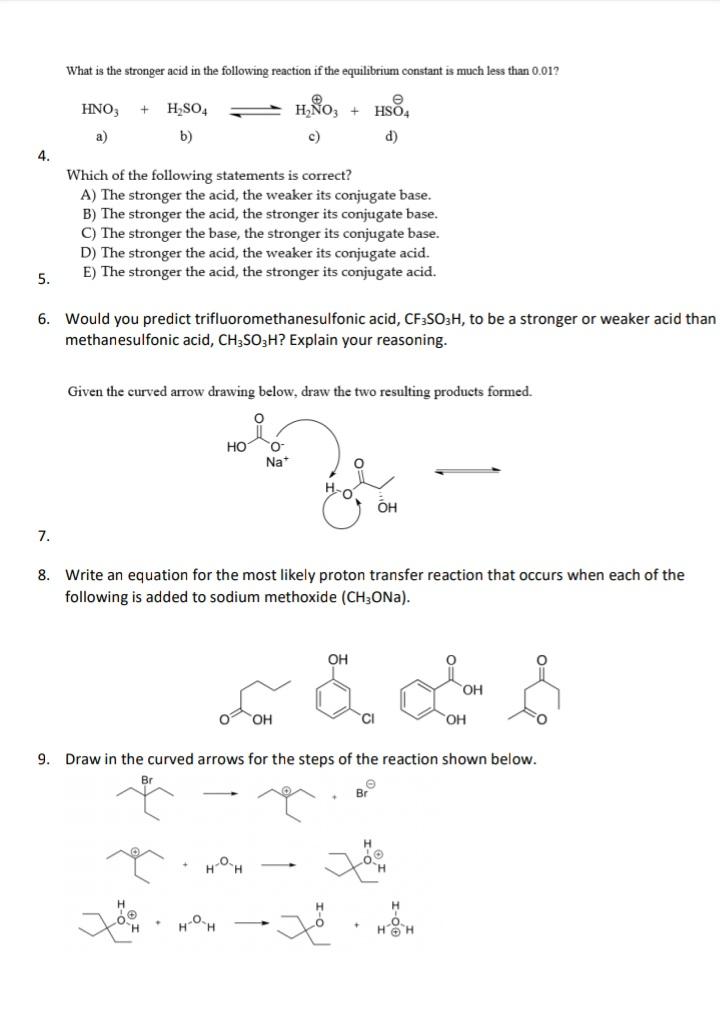
Transcribed Image Text:
What is the stronger acid in the following reaction if the equilibrium constant is much less than 0.01? HNO, H;SO4 HÑO, HSO4 а) b) c) d) 4. Which of the following statements is correct? A) The stronger the acid, the weaker its conjugate base. B) The stronger the acid, the stronger its conjugate base. C) The stronger the base, the stronger its conjugate base. D) The stronger the acid, the weaker its conjugate acid. E) The stronger the acid, the stronger its conjugate acid. 5. 6. Would you predict trifluoromethanesulfonic acid, CF3SO3H, to be a stronger or weaker acid than methanesulfonic acid, CH3SO3H? Explain your reasoning. Given the curved arrow drawing below, draw the two resulting products formed. Но O. Na* OH 7. 8. Write an equation for the most likely proton transfer reaction that occurs when each of the following is added to sodium methoxide (CH3ONA). OH HO, OH OH 9. Draw in the curved arrows for the steps of the reaction shown below. H-OH What is the stronger acid in the following reaction if the equilibrium constant is much less than 0.01? HNO, H;SO4 HÑO, HSO4 а) b) c) d) 4. Which of the following statements is correct? A) The stronger the acid, the weaker its conjugate base. B) The stronger the acid, the stronger its conjugate base. C) The stronger the base, the stronger its conjugate base. D) The stronger the acid, the weaker its conjugate acid. E) The stronger the acid, the stronger its conjugate acid. 5. 6. Would you predict trifluoromethanesulfonic acid, CF3SO3H, to be a stronger or weaker acid than methanesulfonic acid, CH3SO3H? Explain your reasoning. Given the curved arrow drawing below, draw the two resulting products formed. Но O. Na* OH 7. 8. Write an equation for the most likely proton transfer reaction that occurs when each of the following is added to sodium methoxide (CH3ONA). OH HO, OH OH 9. Draw in the curved arrows for the steps of the reaction shown below. H-OH
Expert Answer:
Answer rating: 100% (QA)
3 As we know sulphuric acid is stronger acid than nitric acid su... View the full answer

Related Book For 

Auditing and Assurance Services
ISBN: 978-0077862343
6th edition
Authors: Timothy Louwers, Robert Ramsay, David Sinason, Jerry Straws
Posted Date:
Students also viewed these chemistry questions
-
Which is the stronger acid in each of the following pairs? Explain your reasoning. (a) Phenol or p-hydroxybenzaldehyde (b) m-Cyanophenol or p-cyanophenol (c) o-Fluorophenol or p-fluorophenol
-
Which of the following statements is CORRECT? O a. Stockholders in general would be better off if managers never disclosed favorable events and therefore caused the price of the firm's stock to sell...
-
Write an equation for proton transfer from hydrogen chloride to tert-butyl alcohol. Use curved arrows to track electron movement, and identify the acid, base, conjugate acid, and conjugate base.
-
15. The temperature C, in degrees, that is equivalent to a temperature of F degrees Fahrenheit is given by C- graph of this equation shows the temperature in Celsius for the corresponding...
-
An invoice for $3200, dated March 20, terms 3/10 E.O.M., was received March 23. What payment must be made on April 10 to reduce the debt to $1200?
-
Explain the Internet of Things (IoT) and Blockchain.
-
Construct a relative frequency histogram using the frequency distribution in Exercise 17. Then determine which class has the greatest relative frequency and which has the least relative frequency....
-
Briefly describe the elements of a business case for a new information systems project under the SDLC methodology.
-
If the balance of Work in Process on August 31 is $220,000, what was the amount debited to Work in Process for factory overhead in August, assuming a factory overhead rate of 30% of direct labor...
-
Naboni was established in 1996 by Mr. Nicholas Menyani following a decision by the government of Zambia to liberalize the Zambian economy. Prior to 1991, the Zambian economy had been dominated by...
-
What communication strategies are associated with effective leadership? Explain
-
Apply the marketing-myopia concept to print media, magazines, and newspapers. What is the implication?
-
Consider the Gallo strategic decision. Describe how you would go about evaluating that decision.
-
In question 1, identify the marketing as opposed to the business strategy. Data From Question 1: What is a business strategy? Do you agree with the definition proposed? Illustrate your answer with...
-
Pick an industry and a product or service. Engage in a creative-thinking process, as outlined in Chapter 11, to generate an improved offering. Do the same to create an entirely new offering that uses...
-
Pick a product like Cadillac or Sara Lee Deli products or service like Mr. Clean Performance Car Wash or a car and home insurance company that is offered in a limited number of countries. Assess the...
-
Discuss some of the characteristics of a NoSQL database. Include specific examples of popular online companies, such as Amazon; which uses NoSQL databases.
-
The baseball player A hits the ball from a height of 3.36 ft with an initial velocity of 34.8 ft/s. 0.14 seconds after the ball is hit, player B who is standing 15 ft away from home plate begins to...
-
What roles must a professional accountant be prepared to perform in regard to ethical decision problems?
-
What requirements are usually necessary to become licensed as a certified public accountant? a. Successful completion of the Uniform CPA Examination. b. Experience in the accounting field. c....
-
An environmental audit might include all of following except a. Determining that proper tracking of waste material is being maintained by the organization. b. Reviewing the liability account...
-
What is the impact of the Internet on international business? Which companies and which countries will gain as Internet usage increases throughout the world? Which will lose?
-
How do merchandise exports and imports differ from service exports and imports?
-
What is portfolio investment?

Study smarter with the SolutionInn App


