A) Determine Activation energy using The Arrhenius Equation: k=Ae^-Ea/RT use Rate Law to help solve for k.
Fantastic news! We've Found the answer you've been seeking!
Question:
A) Determine Activation energy using The Arrhenius Equation: k=Ae^-Ea/RT
use Rate Law to help solve for k.
Rate Law=k[I-][H2O2]
B) Use info above to create a 6 plot include a trend line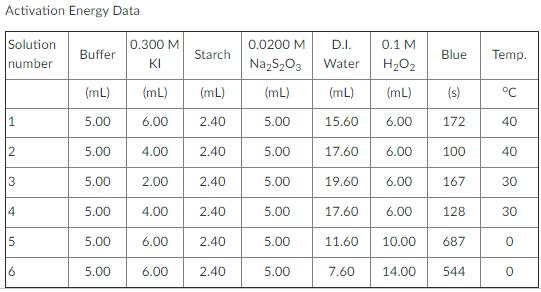
Related Book For 

Posted Date:




