Question: After addressing the Project Engineer's concerns, he then shows you a map of the local region prepared by the contracted geologist, whose contract has
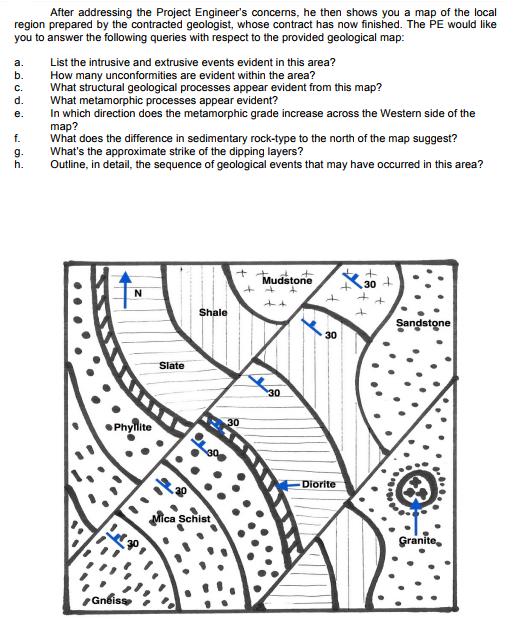
After addressing the Project Engineer's concerns, he then shows you a map of the local region prepared by the contracted geologist, whose contract has now finished. The PE would like you to answer the following queries with respect to the provided geological map: a. List the intrusive and extrusive events evident in this area? How many unconformities are evident within the area? What structural geological processes appear evident from this map? b. C. d. What metamorphic processes appear evident? In which direction does the metamorphic grade increase across the Western side of the map? What does the difference in sedimentary rock-type to the north of the map suggest? What's the approximate strike of the dipping layers? Outline, in detail, the sequence of geological events that may have occurred in this area? . f. g. h. Mudstone 30 N Shale Sandstone 30 Slate 30 Phyflite 30 Diorite Mica Schist Granite, Gneiss
Step by Step Solution
3.42 Rating (161 Votes )
There are 3 Steps involved in it
1 There are just two nosy occasions for example Interruption of diorite ledge and interruption of ro... View full answer

Get step-by-step solutions from verified subject matter experts


