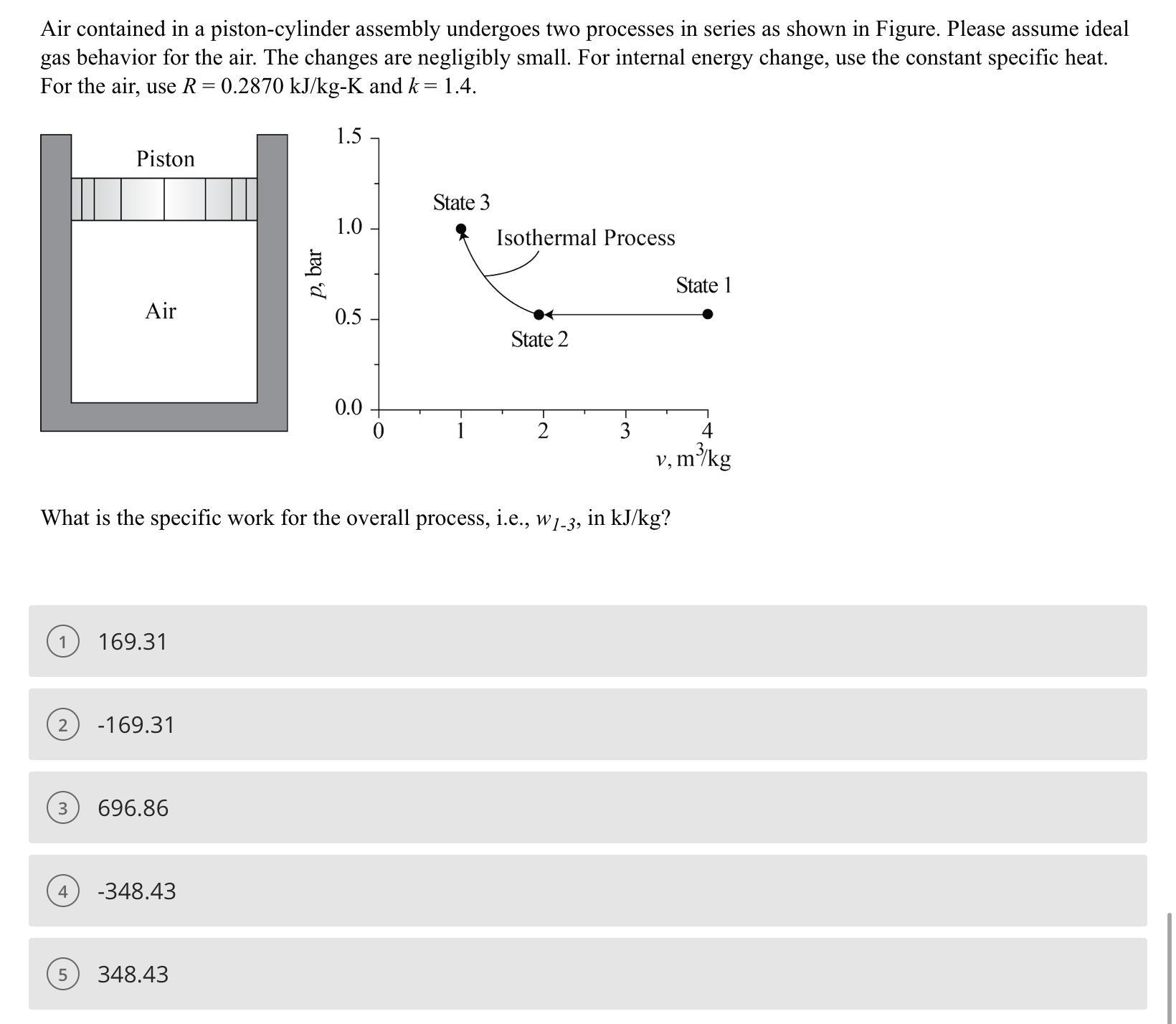
Air contained in a piston-cylinder assembly undergoes two processes in series as shown in Figure. Please...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Air contained in a piston-cylinder assembly undergoes two processes in series as shown in Figure. Please assume ideal gas behavior for the air. The changes are negligibly small. For internal energy change, use the constant specific heat. For the air, use R = 0.2870 kJ/kg-K and k = 1.4. Piston 3 Air 4 2 -169.31 169.31 696.86 -348.43 p, bar 5 348.43 1.5 1.0 0.5 What is the specific work for the overall process, i.e., w1-3, in kJ/kg? 0.0 State 3 Isothermal Process State 2 3 State 1 v, m³/kg Air contained in a piston-cylinder assembly undergoes two processes in series as shown in Figure. Please assume ideal gas behavior for the air. The changes are negligibly small. For internal energy change, use the constant specific heat. For the air, use R = 0.2870 kJ/kg-K and k = 1.4. Piston 3 Air 4 2 -169.31 169.31 696.86 -348.43 p, bar 5 348.43 1.5 1.0 0.5 What is the specific work for the overall process, i.e., w1-3, in kJ/kg? 0.0 State 3 Isothermal Process State 2 3 State 1 v, m³/kg
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Sure based on the information in the im... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these finance questions
-
The small cylinder C has as mass of 10 kg and is attached to the end of a rod whose mass may be neglected. If the frame is subjected to a couple M = (8t2 + 5) N, m where t is in seconds, and the...
-
Pulses A, B, C, and D all travel at 10 m>s on the same string but in opposite directions. The string is depicted at time t = 0 in FIGURE 14-45. The small pulses have an amplitude of 2.0 cm, and the...
-
The furnace cover has a mass of 20 kg and a radius of gyration k G = 0.25 m about its mass center G. If an operator applies a force F = 120 N to the handle in order to open the cover, determine the...
-
(a) Find the Maclaurin series for the function f(x)= ln(1+x) and hence that for In(1+x) (b) By keeping the first four terms in the Maclaurin series for In(1+x) integrate the function In(1+x) from x =...
-
Lukow Products is investigating the purchase of a piece of automated equipment that will save $400,000 each year in direct labor and inventory carrying costs. This equipment costs $2,500,000 and is...
-
To what types of assets does amortization expense apply under ASPE?
-
Identify the research scenario, including the general area of focus. Develop a hypothetical research scenario that would necessitate the use of the Mixed Method A-B-A Design. The research will be...
-
Being a prudent investor, Sally Perkins always investigates a company thoroughly before purchasing shares of its stock for investment. Ms. Perkins is interested in the common stock of Plunge...
-
How can we add database connection script to store form input values into MySQL Database Table such as LogIn tables.
-
Use the area formula (4-- bh) to find the area of each triangle. Note that the base and height are labeled for you. (Watch units! There are 3 feet in a yard and 10 mm in a cm.) 29) 30) 8 yd 7.7 yd 11...
-
How many times is the text "Let us C" printed if the code snippet given below is run? int i = 0; do { cout < < "Let us C" < < endl; i++; if (i 2 == 0) { i = 11; } } while (i
-
4) A proposed new investment has projected sales of $515,000. Variable costs are 37 percent of sales, and fixed costs are $133,000; depreciation is $52,000. Prepare a pro forma income statement...
-
You have been asked to build a web application for your local soccer club which will record and display fixtures for matches. A match fixture should contain information about the competition (such as...
-
A pendulum makes 42 cycles in 50.0 seconds. What is its period? What is its frequency?
-
Can you elucidate the role of strategic foresight and horizon scanning in long-term planning processes, including the identification of emerging trends, disruptive technologies, and market...
-
A tank contains methane 30 C and 5 atm of pressure. What is the internal pressure when heated to 90 C?
-
The area of square PQRS is 100 ft2, and A, B, C, and D are the midpoints of the sides. Find the area of square ABCD. B A
-
Seawater containing 3.50 wt % salts passes through a series of 10 evaporators. Roughly equal quantities of water are vaporized in each of the 10 units and then condensed and combined to obtain a...
-
Ammonia is oxidized with air to form nitric oxide in the first step of the production of nitric acid. Two principal reactions occur: 4 NH 3 + 5O 2 ? 4NO + 6H 2 O 2 NH 3? + 3/2 O 2? ? N 2? + 3H 2 O A...
-
Perform the following estimations without using ca calculator. (a) Estimate the mass of water (kg) in an Olympic-size swimming pool. (b) A drinking glass is being filled from a pitcher. Estimate the...
-
In defining asset classes as part of the strategic asset allocation decision, pairwise correlations within asset classes should generally be: A. Equal to correlations among asset classes. B. Lower...
-
Tactical asset allocation is best described as: A. Attempts to exploit arbitrage possibilities among asset classes. B. The decision to deliberately deviate from the policy portfolio. C. Selecting...
-
The timing of payouts for property and casualty insurers is unpredictable (lumpy) in comparison with the timing of payouts for life insurance companies. Therefore, in general, property and casualty...

Study smarter with the SolutionInn App


