Alake Company is a manufacturing firm that uses job-order costing. At the beginning and ending of...
Fantastic news! We've Found the answer you've been seeking!
Question:
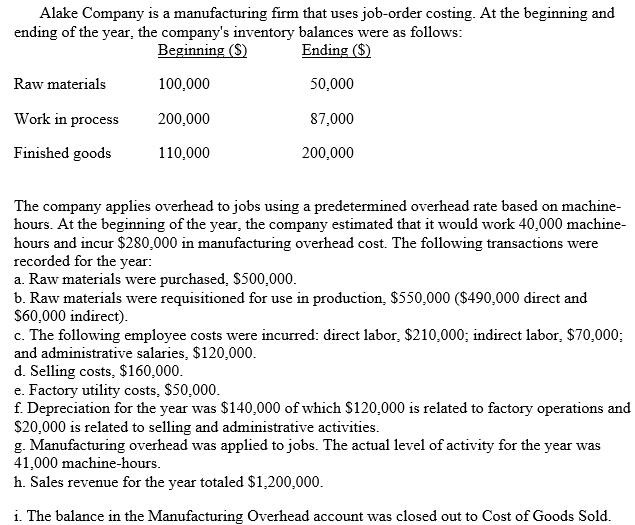

Transcribed Image Text:
Alake Company is a manufacturing firm that uses job-order costing. At the beginning and ending of the year, the company's inventory balances were as follows: Beginning (S) Ending ($) 100,000 50,000 200,000 87,000 110,000 200,000 Raw materials Work in process Finished goods The company applies overhead to jobs using a predetermined overhead rate based on machine- hours. At the beginning of the year, the company estimated that it would work 40,000 machine- hours and incur $280,000 in manufacturing overhead cost. The following transactions were recorded for the year: a. Raw materials were purchased, $500,000. b. Raw materials were requisitioned for use in production, $550,000 ($490,000 direct and $60,000 indirect). c. The following employee costs were incurred: direct labor, $210,000; indirect labor, $70,000; and administrative salaries, $120,000. d. Selling costs, $160,000. e. Factory utility costs, $50,000. f. Depreciation for the year was $140,000 of which $120,000 is related to factory operations and $20,000 is related to selling and administrative activities. g. Manufacturing overhead was applied to jobs. The actual level of activity for the year was 41,000 machine-hours. h. Sales revenue for the year totaled $1,200,000. i. The balance in the Manufacturing Overhead account was closed out to Cost of Goods Sold. Required: 1. Compute a. Costs of goods manufactured for the b. Costs of goods sold for the year, c. Net income for the year. 2. Prepare any necessary journal entries for the costs of goods manufactured and costs of goods sold. 3. Prepare the appropriate journal entry for each of the items above (a. through i.). You can assume that all transactions with employees, customers, and suppliers were conducted in cash. year, Alake Company is a manufacturing firm that uses job-order costing. At the beginning and ending of the year, the company's inventory balances were as follows: Beginning (S) Ending ($) 100,000 50,000 200,000 87,000 110,000 200,000 Raw materials Work in process Finished goods The company applies overhead to jobs using a predetermined overhead rate based on machine- hours. At the beginning of the year, the company estimated that it would work 40,000 machine- hours and incur $280,000 in manufacturing overhead cost. The following transactions were recorded for the year: a. Raw materials were purchased, $500,000. b. Raw materials were requisitioned for use in production, $550,000 ($490,000 direct and $60,000 indirect). c. The following employee costs were incurred: direct labor, $210,000; indirect labor, $70,000; and administrative salaries, $120,000. d. Selling costs, $160,000. e. Factory utility costs, $50,000. f. Depreciation for the year was $140,000 of which $120,000 is related to factory operations and $20,000 is related to selling and administrative activities. g. Manufacturing overhead was applied to jobs. The actual level of activity for the year was 41,000 machine-hours. h. Sales revenue for the year totaled $1,200,000. i. The balance in the Manufacturing Overhead account was closed out to Cost of Goods Sold. Required: 1. Compute a. Costs of goods manufactured for the b. Costs of goods sold for the year, c. Net income for the year. 2. Prepare any necessary journal entries for the costs of goods manufactured and costs of goods sold. 3. Prepare the appropriate journal entry for each of the items above (a. through i.). You can assume that all transactions with employees, customers, and suppliers were conducted in cash. year,
Expert Answer:
Answer rating: 100% (QA)
Lets calculate the required figures and prepare the necessary journal entries for Alake Company 1 Compute a Cost of Goods Manufactured COGM for the ye... View the full answer

Related Book For 

Managerial Accounting
ISBN: 9780073526706
12th Edition
Authors: Ray H. Garrison, Eric W. Noreen, Peter C. Brewer
Posted Date:
Students also viewed these accounting questions
-
Comparative financial statements for Weller Corporation, a merchandising company, for the year ending December 31 appear below. The company did not issue any common stock during the year. A total of...
-
Two ultrasonic sensors have been mounted on the metal ring. Whilst the view angle between the sensors is X, the beam width is 21 degree. There are three possibilities histograms can be obtained in...
-
Alam Company is a manufacturing firm that uses job-order costing. At the beginning of the year, the company's inventory balances were as follows: Raw materials $ 25,400 Work in process $ 74,400...
-
The inequality describes the range of monthly average temperatures T in degrees Fahrenheit at a certain location. (a) Solve the inequality. (b) If the high and low monthly average temperatures...
-
Write a sequence of steps depicting the mechanisms of each of the following reactions:
-
Hayward Industries manufactures dining chairs and tables. The following information is available: Perform the following analyses for these two components of overhead: Compute total machine setups and...
-
For an automobile moving along a highway, describe the control volume you would use to estimate the flowrate of air across the radiator. Explain how you would estimate the velocity of that air.
-
A thin beam of white light is directed at a flat sheet of silicate flint glass at an angle of 20.0? to the surface of the sheet. Due to dispersion in the glass, the beam is spread out as shown in a...
-
On January 1, 2020, Everly Bottle Company sold $4,000,000 in long-term bonds. The bonds will mature in 4 years and have a stated interest rate of 10% and a yield rate of 8%. The bonds pay interest...
-
Carrie DLake, Reed A. Green, and Doug A. Divot share a passion for golf and decide to go into the golf club manufacturing business together. On January 2, 2020, DLake, Green, and Divot form the...
-
Angela noticed a list of creditors at the bottom of her credit report. Some of the inquiries were voluntary and some were involuntary. What is the difference between the two? Question content area...
-
Objective Move event handler binding from constructor code to event tables Instructions Up until now all events have been configured using the this->Bind method. This is useful, or even necessary, in...
-
Company has 2 million revenue for the old credit policy 1/10 net 30, new credit policy 6/10 net 50 is expected to reduce the price for 5% which will increase the sales for cash for 15% and credit for...
-
Kota Company furnishes you with the following production data for the month of March 2022: The application of materials for department L are as follows: 60% at the start of the process; 20% when the...
-
This project is about preparing a financial ration analysis report about 2 companies over a 5 years timespan. Please use the most recent financial statements like between 2022 and 2017. If you cannot...
-
Price reduction for new credit policy from 1/10 net 30 to 6/10 net 45, how to calculation genertated revenue?
-
48. A 24-year-old G2P1001 woman is at 34 weeks' gestation and noted to be icteric. She also has nausea and vomiting and malaise. A diagnosis of acute fatty liver of pregnancy is made, and the...
-
Walker, Inc., is an all-equity firm. The cost of the company's equity is currently 11.4 percent and the risk-free.rate is 3.3 percent. The company is currently considering a project that will cost...
-
Information on four investment proposals is given below: Required: 1. Compute the project profitability index for each investment proposal. 2. Rank the proposals in terms ofpreference. nvestment...
-
Name three criteria that should be considered in choosing an activity base on which to construct a flexible budget.
-
What is the purpose of a statement of cash flows?
-
The first three energy levels \(E(\mathrm{eV})\) of the fictitious element \(\mathrm{X}\) are \(\ldots . . \ldots\) shown in Figure P29.56. a. What wavelengths are observed in the absorption spectrum...
-
A sodium atom in the \(3 p\) state emits a photon, decreasing the energy of the atom by \(2.10 \mathrm{eV}\). By how much does the orbital angular momentum of the atom change? Does it increase or...
-
A 1.5 V D-cell battery is rated at \(15,000 \mathrm{~mA} \cdot \mathrm{h}\). For how long could such a battery power a flashlight bulb rated at \(1.5 \mathrm{~W}\) ?

Study smarter with the SolutionInn App


