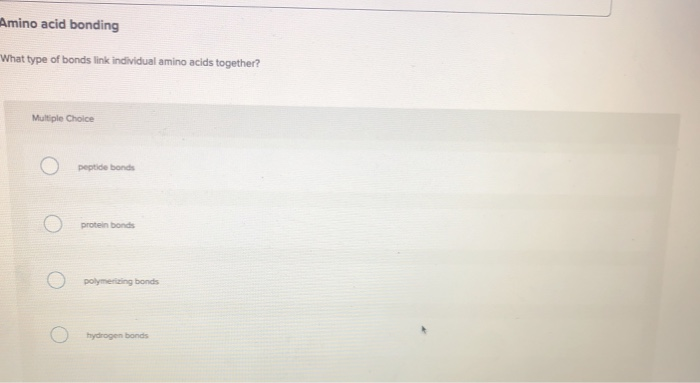
Amino acid bonding What type of bonds link individual amino acids together? Multiple Choice Opeptide bonds...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
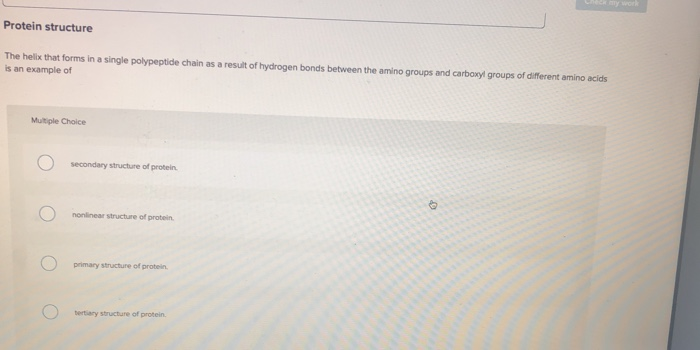
Amino acid bonding What type of bonds link individual amino acids together? Multiple Choice Opeptide bonds protein bonds Opolymerizing bonds hydrogen bonds Protein structure The helix that forms in a single polypeptide chain as a result of hydrogen bonds between the amino groups and carboxyl groups of different amino acids is an example of Multiple Choice secondary structure of protein. nonlinear structure of protein. primary structure of protein. tertiary structure of protein. O Amino acid bonding What type of bonds link individual amino acids together? Multiple Choice Opeptide bonds protein bonds Opolymerizing bonds hydrogen bonds Protein structure The helix that forms in a single polypeptide chain as a result of hydrogen bonds between the amino groups and carboxyl groups of different amino acids is an example of Multiple Choice secondary structure of protein. nonlinear structure of protein. primary structure of protein. tertiary structure of protein. O
Expert Answer:
Answer rating: 100% (QA)
Amino acid bonding peptide bonds Peptide bonds link individual aminoacids together Ex... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The first amino acid incorporated into a polypeptide chain during its biosynthesis in prokaryotes is N-formylmethionine. Explain the purpose of the formyl group.
-
Amino acid compositions can be determined by heating a protein in 6 M HCl and running the hydrolysate through an ion-exchange column. If you were going to do an amino acid sequencing experiment, why...
-
Hydrogen bonding of substrates to enzymes often involves the polypeptide backbone rather than amino acid side chains. What backbone-substrate hydrogen bond helps distort NAM in the D subsite of...
-
Draw a standard normal probability function and show the area under the curve for (a) Plus or minus one standard deviation from the mean (b) Plus or minus two standard deviations from the mean (c)...
-
How should an employer determine its annual pension cost? What minimum GASB criteria must it satisfy?
-
Is the market for laptop computers local, national, or global?
-
William Stem filed an action against Gary Braden, seeking to rescind a contract for the sale of an automobile and to obtain the return of the purchase price plus interest. The trial court granted...
-
The Banisco Corporation is negotiating a contract to borrow $300,000 to be repaid in a lump sum at the end of nine years. Interest payments will be made on the loan at the end of each year. The...
-
Under the independent investor test, all the other factors are examined from the perspective of an independent investor. This requires an analysis of the return on equity (ROE) of the...
-
Express x and y in terms of trigonometric ratios of 0. (Express your answer in terms of 0 only.) X = ? y = ? 16 8 X Y
-
Who investigated Arthur Andersen and Enron?
-
What was the accounting firm that made unethical choices in the Enron scandal ?
-
What are various sections/cost centres or departments within your organisation, which are responsible for financial management of their activities and from whom you would need to collect financial...
-
For the following sequence, determine if it is an arithmetic sequence, a geometric sequence, or neither. If it is either arithmetic or geometric, give the next term in the sequence. 4, 6, 10, 16, 26,...
-
How do you balance your professional and personal life? What type of question is this example? both an open question and primary question both an open question and secondary question secondary...
-
a. Use the table of regression results to answer the questions below (1/2 point each): Left-handed Age Age-squared Constant I D.V.= Wages 1,030,250 (53,649) 76,100 (33,126) -2,420.654 (1625.030)...
-
Write the given system without the use of matrices. D) - ()- d (x sin t + 8 (2+ 1)
-
For the synthesis of ammonia the equilibrium constant Kc at 375°C is 1.2. Starting with [H2]0 = 0.76 M, [N2]0 = 0.60 M, and [NH3]0 = 0.48 M, which gases will have increased in concentration and...
-
Which of the following species is not likely to have a tetrahedral shape? (a) SiBr4, (b) NF4+ (c) SF4, (d) BeCl42-, (e) BF4-, (f) AlCl4-
-
Based on your knowledge of the kinetic theory of gases, derive Graham's law [Equation (5.17)].
-
Around $500 billion in U.S. Treasuries are traded each day and many countries hold U.S. Treasuries, as they are considered a very safe investment. For example, as of October 2018, Japan held $1,018.5...
-
You are working at the campus bookstore earning $9.00 per hour. Your manager tells you that in the upcoming year you will get a 2% raise. How does your real wage change if inflation next year is 1%,...
-
In 2018, the federal government spent $4.1 trillion and brought in revenue of $3.3 trillion. a. Is this a budget deficit or surplus, and what is its size? b. If net government debt in 2018 was $15.8...

Study smarter with the SolutionInn App


