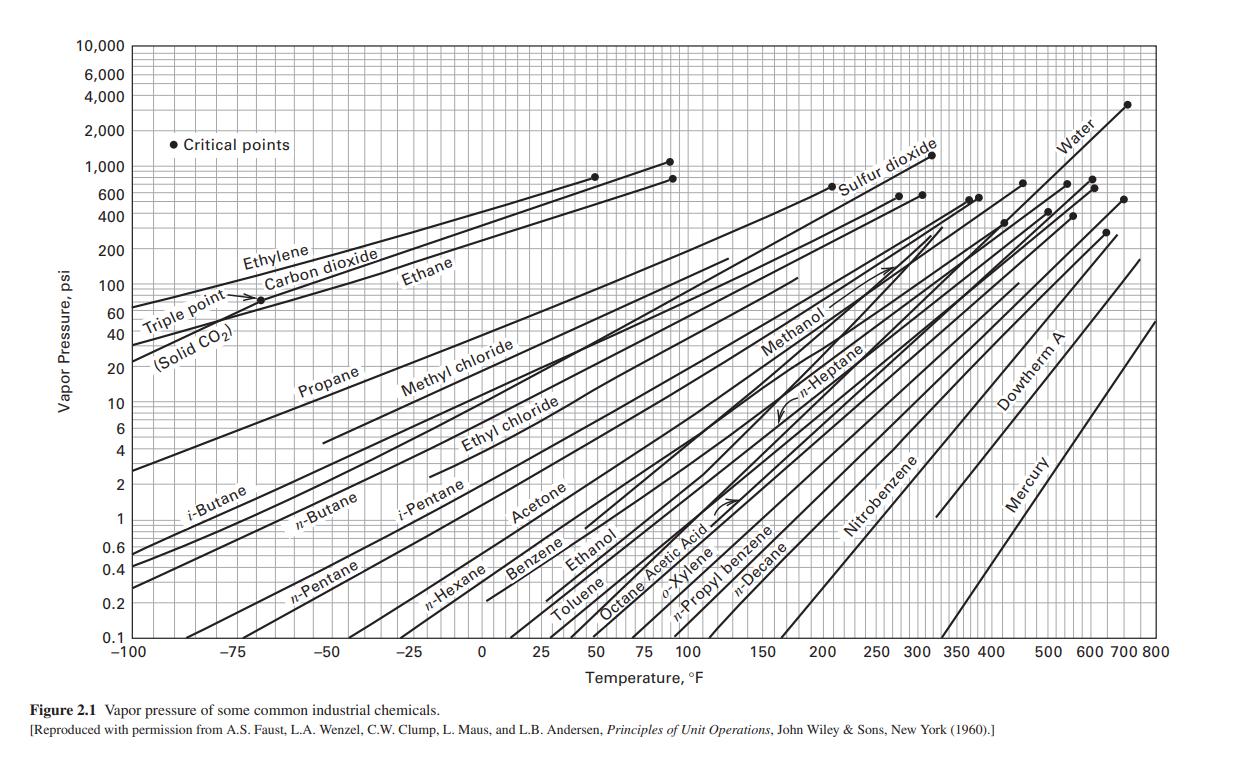
An equimolar solution of benzene and toluene is evaporated at a constant temperature of 90C. What...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
An equimolar solution of benzene and toluene is evaporated at a constant temperature of 90C. What are the pressures at the beginning and end of the vaporization? Assume an ideal solution and use the vapor pressure curves of Figure 2.1, or use a process simulator. Vapor Pressure, psi 10,000 6,000 4,000 2,000 1,000 600 400 200 100 60 40 20 10 6 4 2 1 0.6 0.4 0.2 0.1 Critical points Triple point. (Solid CO) -100 Ethylene. i-Butane -75 Carbon dioxide Propane n-Butane n-Pentane -50 Ethane Methyl chloride Ethyl chloride i-Pentane n-Hexane -25 IN Acetone Ethanol Benzene 0 25 Toluene # Octane Acetic Acid # C 100 50 75 Temperature, F Methanol o-Xylene n-Propyl benzene n-Decane 150 Sulfur dioxide n-Heptane 200 Nitrobenzene 250 300 350 400 Figure 2.1 Vapor pressure of some common industrial chemicals. [Reproduced with permission from A.S. Faust, L.A. Wenzel, C.W. Clump, L. Maus, and L.B. Andersen, Principles of Unit Operations, John Wiley & Sons, New York (1960).] Water therm A Dowtherm A Mercury 500 600 700 800 An equimolar solution of benzene and toluene is evaporated at a constant temperature of 90C. What are the pressures at the beginning and end of the vaporization? Assume an ideal solution and use the vapor pressure curves of Figure 2.1, or use a process simulator. Vapor Pressure, psi 10,000 6,000 4,000 2,000 1,000 600 400 200 100 60 40 20 10 6 4 2 1 0.6 0.4 0.2 0.1 Critical points Triple point. (Solid CO) -100 Ethylene. i-Butane -75 Carbon dioxide Propane n-Butane n-Pentane -50 Ethane Methyl chloride Ethyl chloride i-Pentane n-Hexane -25 IN Acetone Ethanol Benzene 0 25 Toluene # Octane Acetic Acid # C 100 50 75 Temperature, F Methanol o-Xylene n-Propyl benzene n-Decane 150 Sulfur dioxide n-Heptane 200 Nitrobenzene 250 300 350 400 Figure 2.1 Vapor pressure of some common industrial chemicals. [Reproduced with permission from A.S. Faust, L.A. Wenzel, C.W. Clump, L. Maus, and L.B. Andersen, Principles of Unit Operations, John Wiley & Sons, New York (1960).] Water therm A Dowtherm A Mercury 500 600 700 800
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
An equimolar solution of benzene and toluene is totally evaporated at a constant temperature of 90C. What are the pressures at the beginning and end of the vaporization process? Assume an ideal...
-
Benzene and toluene form nearly ideal solutions. Consider an equimolar solution of benzene and toluene. At 20C the vapour pressures of pure benzene and toluene are 9.9 kPa and 2.9 kPa, respectively....
-
Ancient Indians did not believe in war and violence when it came it an expansion of political territory but relied on peaceful negotiation and strategy. Please explain your views on this quote with...
-
Is the GMBG a JIT facility? Why or why not?
-
What is the labor efficiency variance? a. 62,250 unfavorable b. 1,350 favorable c. 63,600 favorable d. 125,850 favorable The following data relate to the budget and actual results of Oscar...
-
Estimate the energy that a runner expends to overcome aerodynamic drag while running a complete marathon race. This expenditure of energy is equivalent to climbing a hill of what height? List all...
-
In the spot market 7.8 pesos can be exchanged for 1 U.S. dollar. A compact disk costs $15 in the United States. If purchasing power parity holds, what should be the price of the same disk in Mexico?
-
ABC; product profitability Outerwear Inc. is concerned about the profitability of its regular gloves. Company managers are considering producing only the top-quality, fleece lined, gloves. The...
-
As of August 31, 20-- Carley's Car Care's general ledger balances, listed in alphabetical order of accounts, are as follows: Accounts Accounts Payable Accounts Receivable Advertising Expense Cash...
-
Why is an increase in accounts receivable as a percentage of sales a signal of improper revenue recognition?
-
A leading sign of an overstatement of sales in Sensormatics fictitious reporting schemes is the increase of accounts receivable as a percentage of sales. True/False
-
When a company sells its accounts receivable, it is important to deduct the amount received on the sale from CFFO before comparing CFFO to operating income when looking for signals of overstatement...
-
Graph the lines in Problems 31-44. \(y=-0.01 x\)
-
To coax even more orders for sales, Sunbeam increasingly gave customers the right to return goods, yet Sunbeam did not increase the _________________ for _________________ in the its accounting...
-
Management Liability is a very unique segment of insurance. Please describe the different lines of insurance: Directors & Officers ; Employment Practices Liability, Fiduciary and Crime. Whar are...
-
What is a content filter? Where is it placed in the network to gain the best result for the organization?
-
It has been shown that a liquid membrane of aqueous ferrous ethylenediaminetetraacetic acid, maintained between two sets of micro porous, hydrophobic, hollow fibers that are packed in a permeator...
-
A reboiled stripper in a natural-gas plant is to be used to remove mainly propane and lighter components from the feed shown in figure. Determine by the group method the compositions of the vapor and...
-
Using the membrane and feed conditions of and values for Rm, and K2 determined in Example 14.15 for DEF microfiltration, compute and plot the permeate flux and cumulative permeate volume as a...
-
How can we effectively analyze a firm's financial ratios?
-
BlackBerry Ltd. has a target current ratio of 2.0 but has experienced some difficulties financing its expanding sales in the past few months. At present, the firm has current assets of $750,000 and a...
-
Cheung Keong Ltd. currently has \($2,150,000\) in current assets and \($860,000\) in current liabilities. The companys managers want to increase the firms inventory, which will be financed by a...

Study smarter with the SolutionInn App


