An ideal gas in a thermally insulated vessel at internal pressure = P, volume = V...
Fantastic news! We've Found the answer you've been seeking!
Question:
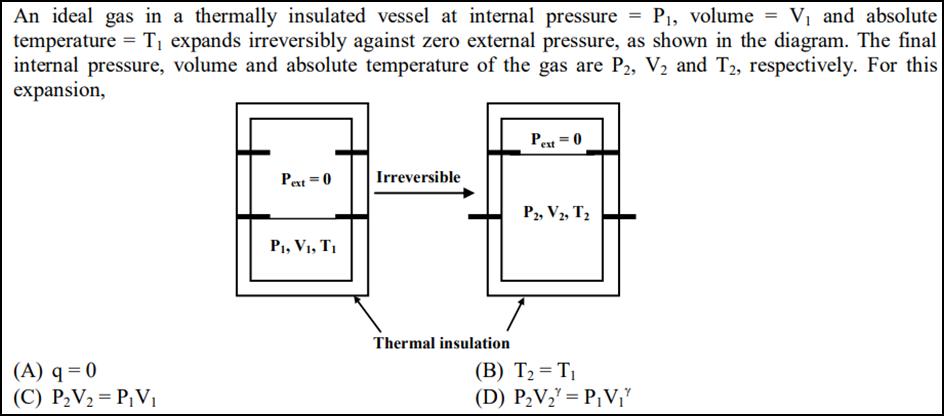
Transcribed Image Text:
An ideal gas in a thermally insulated vessel at internal pressure = P, volume = V and absolute temperature = T expands irreversibly against zero external pressure, as shown in the diagram. The final internal pressure, volume and absolute temperature of the gas are P2, V and T2, respectively. For this expansion, (A) q=0 (C) PV = PV Pext = 0 P, V, T Irreversible Thermal insulation Pext = 0 P2, V2, T (B) T = T (D) PV = PV" An ideal gas in a thermally insulated vessel at internal pressure = P, volume = V and absolute temperature = T expands irreversibly against zero external pressure, as shown in the diagram. The final internal pressure, volume and absolute temperature of the gas are P2, V and T2, respectively. For this expansion, (A) q=0 (C) PV = PV Pext = 0 P, V, T Irreversible Thermal insulation Pext = 0 P2, V2, T (B) T = T (D) PV = PV"
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
14 Thermodynamics and Thermochemistry . The reaction, MgO(s) + C(s) Mg(s) + CO(g ) 18 The entropy change associated with the conversion of 1 kg of ice at 273 K to water vapours at 383 K is for which...
-
A thermally insulated vessel with gaseous nitrogen at a temperature t = 27 C moves with velocity v = 100 m/s. How much (in per cent) and in what way will the gas pressure change on a sudden stoppage...
-
A 1.00-mole sample of an ideal gas in a vessel with a movable piston initially occupies a volume of 5.00 L at an external pressure of 5.00 atm. a. If Pex is suddenly lowered to 2.00 atm and the gas...
-
Explain the purpose of each of the following accounts used in a public share issue: Share Capital, Application, Cash Trust, Allotment, Call, Calls in Advance.
-
Ross Sales had the following transactions for DVDs in 2010, its first year of operations. During the year, Ross Sales sold 850 DVDs for $60 each. Required a. Compute the amount of ending inventory...
-
Information about direct materials cost follows for a local company. Standard price per materials gram ......................................$16 Actual quantity used...
-
If the maximum anticipated hoist load is 12 kip, determine if the W8 \(\times 31\) wide-flange A-36 steel column is adequate for supporting the load. The hoist travels along the bottom flange of the...
-
Stan Fawcetts company is considering producing a gear assembly that it now purchases from Salt Lake Supply, Inc. Salt Lake Supply charges $4 per unit with a minimum order of 3,000 units. Stan...
-
A force acts on a rectangular block as shown. 0.7 m 230 N 39 N 0.5 m 460 N + X4 If the block slides with constant speed, find the position x of the resultant normal force N to the left of the leading...
-
You work in the human resources department of your company helping new employees fill out the necessary paperwork to get their first paycheck. There are a number of decisions that employees must make...
-
In designing a bituminous mixture for a highway surface course to support the traffic loading, data in table below obtained using Marshall method. Determine the optimum asphalt content for 4% VTM?...
-
It takes sunlight about \(8 \mathrm{~min}\) to travel from the Sun to Earth, where it has an average intensity of \(1400 \mathrm{~W} / \mathrm{m}^{2}\). If it takes \(44 \mathrm{~min}\) for light to...
-
A parallel-plate capacitor with circular plates has a steady charging current of \(5.0 \mathrm{~A}\). The wires into and out of the plates attach to the plate centers. If the radius of each plate is...
-
Assume that you have shorted the call option in Problem 2. a. If the stock is trading at $46 in three months, what will you owe? b. If the stock is trading at $32 in three months, what will you owe?...
-
A laser beam has a radius of \(1.5 \mathrm{~mm}\). How powerful does the laser have to be for the maximum magnitude of the magnetic field in the beam to be \(5.0 \mu \mathrm{T}\) ?
-
Full-body scanners at airports are sometimes referred to as millimeter-wave scanners and sometimes as terahertz scanners. Does this make sense?
-
It is around 8.45pm in the Champion Sports Bar which is super busy tonight. There is a small 18 th birthday party in progress (from 7pm until 9pm) and several other groups in the bar. Jim, the...
-
Integration is a vital concept when applied in one?s life. Integrating your life means making ideal choices. Perfect choices on the other go in line with quality decisions. Quality decisions lead to...
-
A student has prepared a cobalt complex that has one of the following three structures: [Co(NH3)6]Cl3, [Co(NH3)5Cl]Cl2, or [Co(NH3)4Cl2]Cl. Explain how the student would distinguish between these...
-
A mixture of Na2CO3 and MgCO3 of mass 7.63 g is reacted with an excess of hydrochloric acid. The CO2 gas generated occupies a volume of 1.67 L at 1.24 atm and 26C. From these data, calculate the...
-
Potash is any potassium mineral that is used for its potassium content. Most of the potash produced in the United States goes into fertilizer. The major sources of potash are potassium chloride (KCl)...
-
Figure P18-39 shows two similar unit pulse waveforms. The only difference is that the first pulse is positive from \(-T / 2\) to \(T / 2\), while the second pulse goes from \(+\mathrm{A}\) to...
-
Identify the essential parts of the definition of auditing.
-
Contrast the activities of independent, internal, and governmental auditors.

Study smarter with the SolutionInn App



