An ideal gas with volume V and pressure P is made to expand adibatically to volume...
Fantastic news! We've Found the answer you've been seeking!
Question:
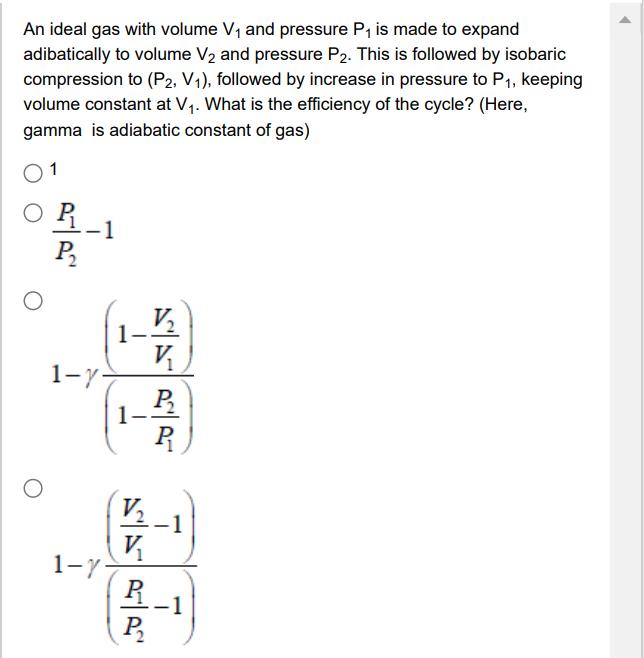
Transcribed Image Text:
An ideal gas with volume V and pressure P is made to expand adibatically to volume V and pressure P2. This is followed by isobaric compression to (P2, V1), followed by increase in pressure to P, keeping volume constant at V. What is the efficiency of the cycle? (Here, gamma is adiabatic constant of gas) 1 P 1-y- 1-7- 1- V V V V R P P R -1 An ideal gas with volume V and pressure P is made to expand adibatically to volume V and pressure P2. This is followed by isobaric compression to (P2, V1), followed by increase in pressure to P, keeping volume constant at V. What is the efficiency of the cycle? (Here, gamma is adiabatic constant of gas) 1 P 1-y- 1-7- 1- V V V V R P P R -1
Expert Answer:
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Posted Date:
Students also viewed these physics questions
-
An ideal gas with constant heat capacities undergoes a change of state from conditions T1, P1 to conditions T2, P2. Determine (H (J mol-1) and (S (J mol-1 K-1) for one of the following cases. (a) T1...
-
An ideal gas with the adiabatic exponent y goes through a process p = Po- aV, where Po and a are positive constants, and V is the volume. At what volume will the gas entropy have the maximum value?
-
An ideal gas with the adiabatic exponent undergoes a process in which its internal energy relates to the volume as U = aV, where a and a are constants. Find: (a) The work performed by the gas and the...
-
A solid of constant density is bounded below by the plane z = 0, on the sides by the elliptical cylinder x 2 + 4y 2 = 4, and above by the plane z = 2 - x. a. Find x and y. b. Evaluate the integral...
-
Plot 1 in Figure a gives the charge q that can be stored on capacitor 1 versus the electric potential 1 set up across it. The vertical scale is set by qs = 16.0C, and the horizontal scale is set by...
-
Evaluate the suitability of M2 as an intermediate target.
-
In the Capital Asset Pricing Model formula, the expected return to the market portfolio, E(R M ), represents the: a. X variable b. Y variable c. Slope d. None of the above
-
The diameter of steel rods manufactured on two different extrusion machines is being investigated. Two random samples of sizes n1 = 15 and n2 = 17 are selected, and the sample means and sample...
-
Orion Iron Corporation tracks the number of units purchased and sold throughout each year but applies its inventory costing method at the end of the year, as if it uses a periodic inventory system....
-
The frame of the roller has a mass of 5.5 Mg and a center of mass at G. The roller has a mass of 2 Mg and a radius of gyration about its mass center of k A = 0.45 m. If a torque of M = 600 N m is...
-
2. Consider a consumer with preferences over current and future consumption given by 1/2 U(C1, C2) (1) (2) 1/2 = where c denotes the amount consumed in period 1 and c the amount consumed in period 2....
-
Explain the two major types of measure used in conventional accounting and ecological accounting. When consideration is given to environmental issues in accounting, what are the two main groups of...
-
Simons Realty issued \(\$ 250,000\) of \(6 \%, 10\)-year bonds payable at par value on May 1,2006, four months after the bond's original issue date of January 1, 2006. Journalize the following...
-
The Xeron Computer Company is considering a design change that would reduce the weight of its high-end desktop computer by 2 kilograms. This design change would cost \($300,000\) to implement for all...
-
You can find the home page for Caterpillar, Inc., at www.caterpillar.com. Go to the web page, select Cat Stock, and find the most recent annual report. Using the information from the financial...
-
A nursery has the choice of planting trees in the ground or in pots prior to sale. The pots cost \($5\) each and potted trees must be watered 100 times per year. Trees that are planted in the ground...
-
Dickson, Inc. has a debt-value ratio of 0.5. The firm's weighted average cost of capital (WACC) is 10 percent, and its pretax cost of debt is 6 percent. The tax rate is 24 percent. What is the...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
If a liquid solution of HCl in water, containing 1 mol of HCl and 4.5 mol of H 2 O, absorbs an additional 1 mol of HCl(g) at a constant temperature of 25C, what is the heat effect?
-
If the heat capacity of a substance is correctly represented by an equation of the form, C P =A+BT+CT 2 show that the error resulting when C P H is assumed equal to C P evaluated at the arithmetic...
-
Consider a binary mixture for which the excess Gibbs energy is given by G E /RT = 2.6x 1 x 2 . For each of the following overall compositions, determine whether one or two liquid phases will be...
-
How can a profitable business fail? a Because it can't pay its bills b Because it has more current liabilities than current assets c Because it has a bank overdraft d Because it has too much cash
-
When adjusting an operating profit to calculate the 'cash flows from operating activities', an increase in inventories over the financial period would be: a Added to operating profit b Ignored c...
-
Cash has been described as: a The lifebuoy of the business b The lifeboat of the business c The lifeline of the business d The lifeblood of the business

Study smarter with the SolutionInn App



