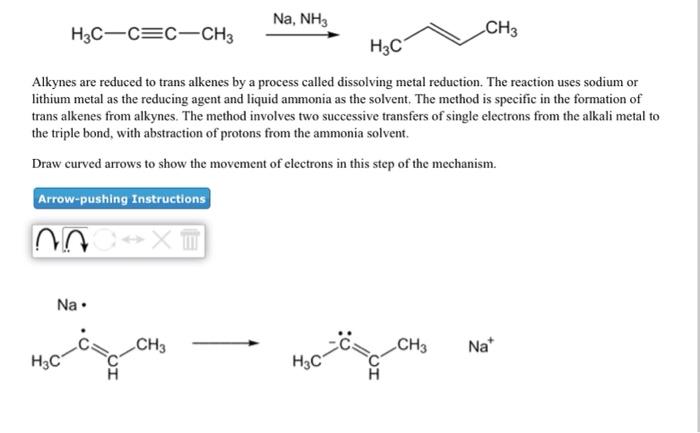
ANO XT H3C-C=C-CH3 Alkynes are reduced to trans alkenes by a process called dissolving metal reduction....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
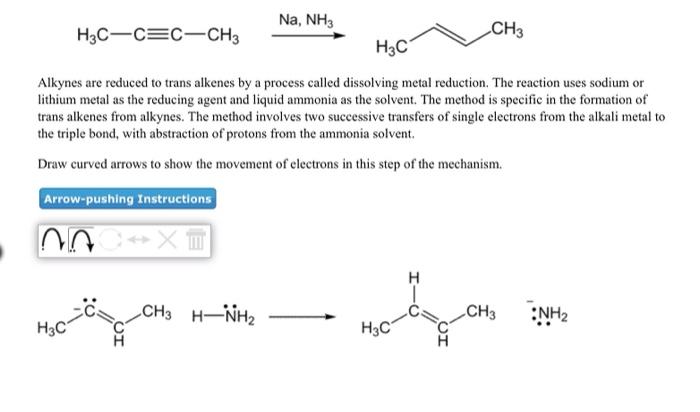
ANO XT H3C-C=C-CH3 Alkynes are reduced to trans alkenes by a process called dissolving metal reduction. The reaction uses sodium or lithium metal as the reducing agent and liquid ammonia as the solvent. The method is specific in the formation of trans alkenes from alkynes. The method involves two successive transfers of single electrons from the alkali metal to the triple bond, with abstraction of protons from the ammonia solvent. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions Na. H3C Na, NH3 CH3 H3C H3C CH3 CH3 Na* H3C Na, NH3 H3C C C CH3 H3C Alkynes are reduced to trans alkenes by a process called dissolving metal reduction. The reaction uses sodium or lithium metal as the reducing agent and liquid ammonia as the solvent. The method is specific in the formation of trans alkenes from alkynes. The method involves two successive transfers of single electrons from the alkali metal to the triple bond, with abstraction of protons from the ammonia solvent. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions NOC XT CH3 HNH2 CH3 40-4₂ CH3 H3C NH₂ ANO XT H3C-C=C-CH3 Alkynes are reduced to trans alkenes by a process called dissolving metal reduction. The reaction uses sodium or lithium metal as the reducing agent and liquid ammonia as the solvent. The method is specific in the formation of trans alkenes from alkynes. The method involves two successive transfers of single electrons from the alkali metal to the triple bond, with abstraction of protons from the ammonia solvent. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions Na. H3C Na, NH3 CH3 H3C H3C CH3 CH3 Na* H3C Na, NH3 H3C C C CH3 H3C Alkynes are reduced to trans alkenes by a process called dissolving metal reduction. The reaction uses sodium or lithium metal as the reducing agent and liquid ammonia as the solvent. The method is specific in the formation of trans alkenes from alkynes. The method involves two successive transfers of single electrons from the alkali metal to the triple bond, with abstraction of protons from the ammonia solvent. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions NOC XT CH3 HNH2 CH3 40-4₂ CH3 H3C NH₂
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Draw curved arrows to show the flow of electrons responsible for the conversion of reactants into products. a. b. c. :O CH-C OCH
-
Draw curved arrows to show the flow of electrons responsible for the conversion of the reactants into the products: Br
-
Use curved arrows to show the movement of electrons in each of the following reaction steps: a. b. c. d. Br O: " CH,COH + H-O-H CH,COH + H2O - CH3 CH3 CH CH3 CH
-
Be able to explain how changes in transportation impacted economic growth in the United States prior to WWI.
-
Which of the following is true according to the pure expectations theory? Forward rates: a. Exclusively represent expected future short rates. b. Are biased estimates of market expectations? c....
-
What is the best approach to address conflict in meetings?
-
The following MINITAB output presents a 95% confidence interval for the mean cholesterol levels for men aged 50 years, and a 95% prediction interval for an individual man aged 50. The units of...
-
The Knowles/Armitage (KA) group at Merrill Lynch advises clients on how to create a diversified investment portfolio. One of the investment alternatives they make available to clients is the All...
-
Write an informative explanation that explores the principles of double - entry accounting, its advantages over single - entry systems, and how it helps maintain accurate financial records for...
-
A corporation owns a chain of several hundred gasoline stations on the eastern seaboard. The marketing director wants to test a proposed marketing campaign by running ads on some local television...
-
Write a paper on Asthma Pathophysiological Mechanism
-
Superior Micro Products uses the weighted - average method in its process costing system. During January, the Delta Assembly Department completed its processing of 2 6 , 2 0 0 units and transferred...
-
A machine was bought on 1 st January 2 0 1 7 for $ 1 2 , 0 0 0 . The policy is to depreciate the machine at 1 0 % on reducing balance method at the end of each year, ending on 3 1 st December....
-
The terminals A and B in figure below are connected to a 9.0-V battery. Suppose that R = 2.7, R2 = 2.5, R3 = 4.8, R = 3.3, R 8.1, and R6 = 6.3. ==
-
A charge of -3.35 nC is placed at the origin of an xy-coordinate system, and a charge of 1.60 nC is placed on the y axis at y = 3.50 cm Part A |F| = K|99| K|99| d If a third charge, of 5.00 nC, is...
-
Consider a previously closed economy that opens up to international trade. Reference the dynamic aggregate demand-aggregate supply framework below to describe a situation where this would lead to...
-
Air at 200 kPa and 400 K is contained in a 10 m insulated tank. The 80 kg weight is lowered by 2 m. The final pressure of air in the tank would be kJ kgK' kPa kJ [c=0.718- R = 0.287- kgk 9= 10 m/s]...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
Several additional amine syntheses are effectively limited to making primary amines. The reduction of azides and nitro compounds and the Gabriel synthesis leave the carbon chain unchanged. Formation...
-
Draw and name all the chlorinated benzenes having from one to six chlorine atoms.
-
Propose structures for intermediates and products (A) through (K). Mg, ether (1) CH CH2CHO (2) H,O l-bromobutane (D) KOH. H2O HzSO, heat (D O3 (2) (CH3.S CH,CH CH CHO + CH3(CH2CHO CH,CH,Br (1) NaNH2,...
-
Derive the equation of motion of Example 4.16 using Newton's second law of motion. Example 4.16 A Rolling Disk The disk on the inclined plane of Figure 4.15 is supported by two massless springs. It...
-
For the problem of oscillator control, given by Equation 4.32, consider the specific governing equation \[ \ddot{x}+2 \zeta \omega_{n} \dot{x}+\omega_{n}^{2} x=\frac{A}{m} \cos \omega t+F_{\text...
-
For Example 4.16 derive the equation of motion using \(x\) as the generalized coordinate. Then solve this equation with initial conditions \(x_{0}, v_{0}\) to find \[ x(t)=\frac{v_{0}}{\omega} \sin...

Study smarter with the SolutionInn App


