What is the pOH of a 0.0250 M HI aqueous solution? For HI, K = 1.0...
Fantastic news! We've Found the answer you've been seeking!
Question:
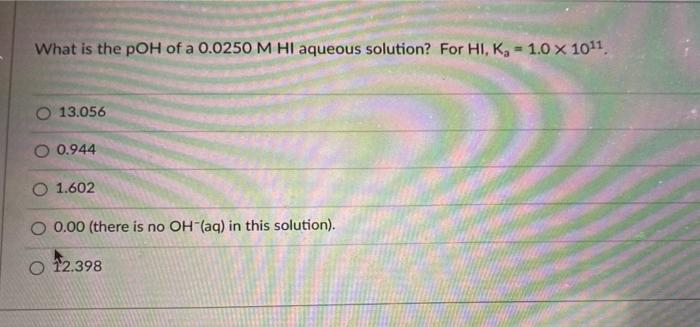

Transcribed Image Text:
What is the pOH of a 0.0250 M HI aqueous solution? For HI, K₂ = 1.0 × 10¹1. O 13.056 O 0.944 O 1.602 O 0.00 (there is no OH(aq) in this solution). O 12.398 Find the pH of a solution that is 0.15 M in HCOOH (K,- 1.8 x 10) and 0.45 M in HCN (K, -4.9 x 1010) at 25°C. O4.83 4.13 3.05 3.87 2.28 What is the pOH of a 0.0250 M HI aqueous solution? For HI, K₂ = 1.0 × 10¹1. O 13.056 O 0.944 O 1.602 O 0.00 (there is no OH(aq) in this solution). O 12.398 Find the pH of a solution that is 0.15 M in HCOOH (K,- 1.8 x 10) and 0.45 M in HCN (K, -4.9 x 1010) at 25°C. O4.83 4.13 3.05 3.87 2.28
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Qu... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Pls solve the problem urgently I will give upvote if you know only. Question Description (a) Calculate the mass attenuation coefficient of silica glass (SiO2, p = 2.21 g/cm3) for 5-MeV y-rays. (b)...
-
Please provide excel with formulas and values both, upvote will be given CASE 4.1 BLENDING AVIATION GASOLINE AT JANSEN GAS ansen Gas creates three types of aviation gaso- line (avgas), labeled A, B,...
-
Please link the article you used (Will upvote if article is published in 2021) Prepare an analysis of a current (published after 1 January 2021) Australian newspaper/media article. ( pick an article...
-
I have this data frame with the years and values, can you make a model to predict the value of meats,fish,fruits and vegetables and grains? based on the current values of the table and historical...
-
Define the different components (y, x, a, and b) of a linear regression equation.
-
The following transactions took place at Sonoma Auto Parts and Custom Shop during the first week of July. Indicate how these transactions would be entered in a purchases journal like the one shown in...
-
Lagos Toy Company reported the following comparative balance sheet: Compute for Lagos: a. Collections from customers during 2009. Sales totaled $140,000. b. Payments for inventory during 2009. Cost...
-
Leisure is an inferior good for Horace. a. Use indifference curves to show the income and substitution effects of an increase in Horaces wage rate. b. Could Horaces labor supply curve be...
-
This is individual assignment. TASK A literature review will require you to source (at least 3) readings relevant to the issues you identified with the business selected. These readings need to be...
-
Lolas operates a chain of sandwich shops. The company is considering two possible expansion plans. Plan A would open eight smaller shops at a cost of $8,550,000. Expected annual net cash inflows are...
-
Mary Marsh, head aerobic instructor at Tuff-it-Out Fitness Centers, wanted to estimate the proportion of all 19-39 year olds who aerobically exercise at least once each week. Mary would like the...
-
Attorney KristinKristin MaloneyMaloney invoiced WilsonWilson for $ 33 comma 450$33,450 and has agreed to accept 2 comma 2002,200 shares of its $ 0.01$0.01 par-value common stock in full payment for...
-
2. Library Research Your college, university, or public library has a wealth of secondary sources for most research topics. You may use the online or card catalogue to find books on your research...
-
What is the approximate uncertainty in the area of a circle of radius 4.1 x 10cm? Express your answer using one significant figure.
-
Julie and Kate form an equal partnership during the current year. Julie contributes cash of $200,000, and Kate contributes property (adjusted basis of $90,000, fair market value of $260,000). The...
-
Smokey and the Bandit produces outdoor activity clothing. The product line consists of pants, jackets, tops, and accessories. Data has been collected related to direct materials and direct labor for...
-
What is the minimum size septic tank for an apartment building with 11 one-bedroom units, 5 two- bedroom units, and 2 three-bedroom units? Hint 1: Table 10.8 in the textbook. Or see below. Hint 2:...
-
The figure shows a bolted lap joint that uses SAE grade 8 bolts. The members are made of cold-drawn AISI 1040 steel. Find the safe tensile shear load F that can be applied to this connection if the...
-
Explain why alkali metals have a greater affinity for electrons than alkaline earth metals.
-
A neutral atom of a certain element has 17 electrons. Without consulting a periodic table, (a) Write the ground-state electron configuration of the element, (b) Classify the element, (c) Determine...
-
Because acid-base and precipitation reactions discussed in this chapter all involve ionic species, their progress can be monitored by measuring the electrical conductance of the solution. Match the...
-
Using Program12.m, find the first five natural frequencies of a thin fixed-fixed beam. Data From Example 8.4:- Find the natural frequencies of a bar with one end fixed and a mass attached at the...
-
Find the first two natural frequencies of a fixed-fixed uniform string of mass density \(ho\) per unit length stretched between \(x=0\) and \(x=l\) with an initial tension \(P\). Assume the...
-
A vehicle, of weight \(F_{0}\), moving at a constant speed on a bridge (Fig. 8.43(a)) can be modeled as a concentrated load traveling on a simply supported beam as shown in Fig. 8.43(b). The...

Study smarter with the SolutionInn App


