Argon gas (10.0 g) is added to a 10.0 L chamber fitted with a piston at...
Fantastic news! We've Found the answer you've been seeking!
Question:
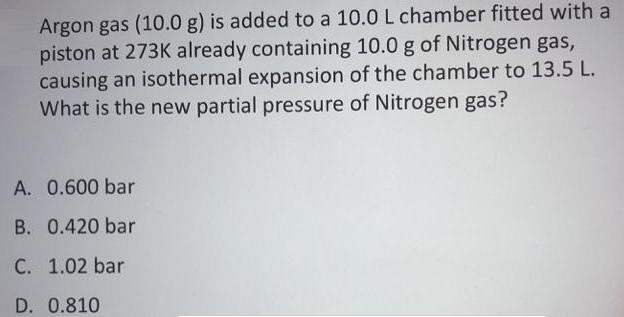
Transcribed Image Text:
Argon gas (10.0 g) is added to a 10.0 L chamber fitted with a piston at 273K already containing 10.0 g of Nitrogen gas, causing an isothermal expansion of the chamber to 13.5 L. What is the new partial pressure of Nitrogen gas? A. 0.600 bar B. 0.420 bar C. 1.02 bar D. 0.810 Argon gas (10.0 g) is added to a 10.0 L chamber fitted with a piston at 273K already containing 10.0 g of Nitrogen gas, causing an isothermal expansion of the chamber to 13.5 L. What is the new partial pressure of Nitrogen gas? A. 0.600 bar B. 0.420 bar C. 1.02 bar D. 0.810
Expert Answer:
Answer rating: 100% (QA)
Moles of argon gas added 10399 where 399 molar mass of Ar Moles of nitrogen 1028 m... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these accounting questions
-
An ideal regenerator (T3 = T5) is added to a simple ideal Brayton cycle (see Fig. P9-99). Air enters the compressor of this cycle at 16 psia and 100°F; the pressure ratio is 11; and the maximum...
-
A 15.9-g sample of sodium carbonate is added to a solution of acetic acid weighing 20.0 g. The two substances react, releasing carbon dioxide gas to the atmosphere. After reaction, the contents of...
-
A 10 mL sample is added to a 300 mL BOD bottle. Dilution water is added to the sample bottle and the initial dissolved-oxygen concentration is measured as 8.5 mg/L. After the sample is sealed, the...
-
You are the assistant vice president in charge of production for a firm that produces computers. Your firm's production function is f(L,K) = min (L,K) Where L and K are the quantities of the two...
-
If the assumption of no market frictions is not true, then why should we study models using this assumption?
-
For a prescribed universe and any open statements p(x), q(x) in the variable x, prove that (a) x [p(x) q(x)] x [p(x) x q(x) (b) x [p(x) q(x) x [p(x) x q(x)
-
Some analysts make a distinction between art and craft (the application of aesthetic principles to the production of utilitarian objects). Discuss and debate whether or not this is a useful...
-
Barbara Flynn is in charge of maintaining hospital supplies at General Hospital. During the past year, the mean lead time demand for bandage BX-5 was 60 (and was normally distributed). Furthermore,...
-
You are trying to install a pool. you need to do some calculations. You will write a Java program named "Circle", that will calculate the area, circumference and volume of the circle. You will need...
-
Is there evidence consistent with gender discrimination in training level? To answer this, proceed as follows: a. Create a table of counts for the two qualitative variables gender and training level....
-
15. An operational amplifier (Op Amp) is a type of electronic circuit that transforms a voltage input, V., to produce a desired output, V. Suppose a particular Op Amp produces a voltage output signal...
-
Internal Evaluation. Internal analysis of the company, to include the analysis of: The company's current situation and the key challenges it needs to overcome (if any) - any existing purpose...
-
How to modify the following algorithms to display the total number of swap operations. The following algorithms are given: def Algorithm_1(lyst): i = 0 while i < len(lyst) - 1: minIndex = i j = i + 1...
-
Propose a NEW operating systems. The NEW operating systems can be based on Windows or Unix/Linux or Mobile OS or independent based platform. Provide details and justifications of your NEW operating...
-
1) What is a material handling system? Explain. 2) List and describe the 5 uses of packaging. 3) Advise on some ways that a company can minimize its potential cargo loss. 4) List 5 types of commonly...
-
DBS Bank is launching a new investment product targeting senior citizens in Asia. The company decides to use direct marketing for selling the product. 1. What direct marketing strategy would allow...
-
AD C+I+G C $200+0.8YD G $1,200 I=$500-30i TA 0.375Y TR=$125 J YD Y TA+TR Ms/P $7,000 and L = Md=2Y-80i The GE solution for the above from your last quiz was: IS Curve: Y $4,000-60i [From Y AD =...
-
Problem 2. (0.6 points, 0.2 points for each question) (a) A company turns its inventory 2 times a month. Its months-of-supply = Its days-of-supply = Please show your analysis below: _months. days. (1...
-
Water in a 5 - cm-deep pan is observed to boil at 98oC. At what temperature will the water in a 40 - cm-deep pan boil? Assume both pans are full of water.
-
Heat is supplied to an absorption refrigeration system from a geothermal well at 110oC at a rate of 5 105 kJ/h. The environment is at 25oC, and the refrigerated space is maintained at - 18oC....
-
A 2.4-m high 200-m2 house is maintained at 22C by an air-conditioning system whose COP is 3.2. It is estimated that the kitchen, bath, and other ventilating fans of the house discharge a houseful of...
-
Natural gas (methane) flows from a 3-in.-diameter gas main, through a 1-in.-diameter pipe, and into the burner of a furnace at a rate of \(100 \mathrm{ft}^{3} /\) hour. Determine the pressure in the...
-
Calculate the Reynolds numbers for the flow of water and for air through a 4-mm-diameter tube, if the mean velocity is \(3 \mathrm{~m} / \mathrm{s}\) and the temperature is \(30^{\circ} \mathrm{C}\)...
-
SAE 30 oil at \(60^{\circ} \mathrm{F}\) flows through a 2-in.-diameter pipe with a mean velocity of \(5 \mathrm{ft} / \mathrm{s}\). Determine the value of the Reynolds number (see Example 1.4)....

Study smarter with the SolutionInn App


