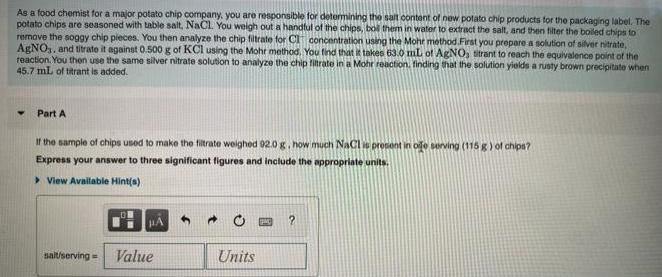
As a food chemist for a major potato chip company, you are responsible for determining the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
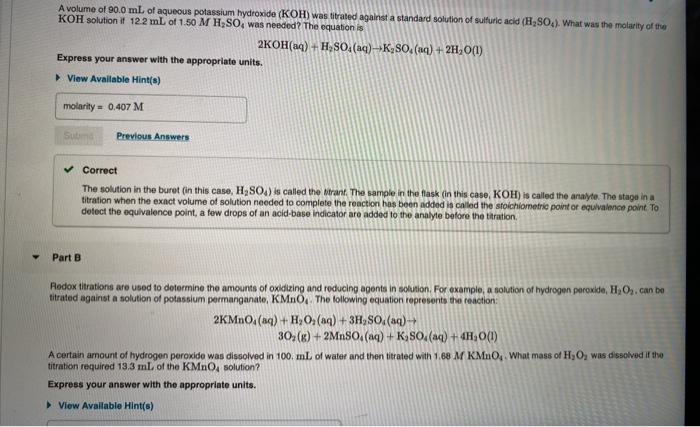
As a food chemist for a major potato chip company, you are responsible for determining the salt content of new potato chip products for the packaging label. The potato chips are seasoned with table salt, NaCl. You weigh out a handful of the chips, bol them in water to extract the salt, and then filter the boiled chips to remove the soggy chip pieces. You then analyze the chip filtrate for CI concentration using the Mohr method.First you prepare a solution of silver nitrate, AgNO,, and titrate it against 0.500 g of KCI using the Mohr method. You tind that it takes 63.0 mL of AgNO, titrant to reach the equivalence point of the reaction. You then use the same silver nitrate solution to analyze the chip filtrate in a Mohr reaction. finding that the solution yields a rusty brown precipitate when 45.7 mL of titrant is added. Part A If the sample of chips used to make the filtrate weighed 92.0 g, how much NaCl is present in ole serving (115 g) of chips? Express your answer to three significant figures and include the appropriate units. > View Available Hint(s) sait/serving = Value Units A volume of 90.0 mL of aqueous potassium hydroxide (KOH) was titrated against a standard solution of sulfuric acid (H2SCO). What was the molarity of the KOH solution if 122 mL of 1.50 M H;SO, was needed? The equation is 2KOH(aq) + H, SO,(aq)-→K2SO, (aq) + 2H,O(1) Express your answer with the appropriate units. > View Available Hint(s) molarity = 0.407 M Sub Previous Answers Correct The solution in the buret (in this case, H2SO4) is called the trant. The sample in the flask (in this case, KOH) is called the analyte. The stage in a titration when the exact volume of solution needed to complete the reaction has been added is called the stoichiometric point or equivalence point. To detect the equivalence point, a few drops of an acid-base indicator are added to the analyte before the titration. Part B Rodox titrations are used to determine the amounts of oxidizing and reducing agents in solution. For example, a solution of hydrogen peroxide, HO2. can be titrated against a solution of potassium permanganate, KMNO The folowing equation represents the reaction: 2KMNO, (aq) + H, O, (aq) + 3H, SO. (aq)- 30; (K) + 2MNSO. (aq) + K,SO.(aq) + 4H, 0(1) A certain amount of hydrogen peroxide was dissolved in 100. mL of water and then titrated with 1.68 M KMnO. What mass of H,O, was dissolved if the titration required 13.3 ml. of the KMNO, solution? Express your answer with the appropriate units. > View Available Hint(s) As a food chemist for a major potato chip company, you are responsible for determining the salt content of new potato chip products for the packaging label. The potato chips are seasoned with table salt, NaCl. You weigh out a handful of the chips, bol them in water to extract the salt, and then filter the boiled chips to remove the soggy chip pieces. You then analyze the chip filtrate for CI concentration using the Mohr method.First you prepare a solution of silver nitrate, AgNO,, and titrate it against 0.500 g of KCI using the Mohr method. You tind that it takes 63.0 mL of AgNO, titrant to reach the equivalence point of the reaction. You then use the same silver nitrate solution to analyze the chip filtrate in a Mohr reaction. finding that the solution yields a rusty brown precipitate when 45.7 mL of titrant is added. Part A If the sample of chips used to make the filtrate weighed 92.0 g, how much NaCl is present in ole serving (115 g) of chips? Express your answer to three significant figures and include the appropriate units. > View Available Hint(s) sait/serving = Value Units A volume of 90.0 mL of aqueous potassium hydroxide (KOH) was titrated against a standard solution of sulfuric acid (H2SCO). What was the molarity of the KOH solution if 122 mL of 1.50 M H;SO, was needed? The equation is 2KOH(aq) + H, SO,(aq)-→K2SO, (aq) + 2H,O(1) Express your answer with the appropriate units. > View Available Hint(s) molarity = 0.407 M Sub Previous Answers Correct The solution in the buret (in this case, H2SO4) is called the trant. The sample in the flask (in this case, KOH) is called the analyte. The stage in a titration when the exact volume of solution needed to complete the reaction has been added is called the stoichiometric point or equivalence point. To detect the equivalence point, a few drops of an acid-base indicator are added to the analyte before the titration. Part B Rodox titrations are used to determine the amounts of oxidizing and reducing agents in solution. For example, a solution of hydrogen peroxide, HO2. can be titrated against a solution of potassium permanganate, KMNO The folowing equation represents the reaction: 2KMNO, (aq) + H, O, (aq) + 3H, SO. (aq)- 30; (K) + 2MNSO. (aq) + K,SO.(aq) + 4H, 0(1) A certain amount of hydrogen peroxide was dissolved in 100. mL of water and then titrated with 1.68 M KMnO. What mass of H,O, was dissolved if the titration required 13.3 ml. of the KMNO, solution? Express your answer with the appropriate units. > View Available Hint(s)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The Crunchy Potato Chip Company sells chips in boxes with a net weight of 30 ounces per box (850 grams). Each box contains ten individual 3-ounce packets of chips. Product design specifications call...
-
A snack food company produces potato chips on three different production lines. Line 1 produces 20% of the total output, Line 2 produces 30%, and Line 3 produces the balance. Each line is sampled to...
-
It takes 23.77 mL of 0.1505 M HCl to titrate with 15.00 mL of Ca(OH)2. What is the concentration of Ca(OH)2? You will need to write the balanced chemical equation first.
-
Krell Industries has a share price of $22 46 today. If Krell is expected to pay a dividend of S0.83 this year, and its stock price is expected to grow to $24.11 at the end of the year, what is...
-
A subsonic airplane is flying at a 5000-m altitude where the atmospheric conditions are 54 kPa and 256 K. A Pitot static probe measures the difference between the static and stagnation pressures to...
-
Complete the following to determine the restrictions on net position at the government-wide level. Be sure to deduct accrued interest on long-term debt in calculating the December 31, 2023, balance...
-
Does object-oriented technology help or hinder testing?
-
In the following figure, a consumer is initially in equilibrium at point C. The consumers income is $ 400, and the budget line through point C is given by $ 400 $ 100X + $ 200Y. When the consumer is...
-
Lansing Company s current - year income statement and selected balance sheet data at December 3 1 of the current and prior years follow. LANSING COMPANY Income Statement For Current Year Ended...
-
55 Jason Ready, single, attended the University of San Diego during 2015-2019 under the Army ROTC program. Excluding the school expenses covered by his ROTC scholarship, he incurred additional school...
-
You own a local bakery in a small town called Darlington. Poor farmer's harvest means that basic raw materials such as wheat have become increasingly expensive. You expect the price rise to persist....
-
Explain with an example how organizations plan and coordinate demand and supply in a supply chain.
-
The research hypothesizes that companies that emphasize LTSM are more likely to share information and encourage collaborative planning, forecasting, and replenishment (CPFR) across supplier tiers....
-
Debt contracts represent fixed claims against the firm, while an equity contract entitles the shareholder to claim against the residual cash flows of the firm. Consider a small firm with one...
-
what types of incentives could a financial institution offer to the unbanked population?
-
Why is a cross functional team consisting of engineering, manufacturing and supply management personnel a positive when contributing to specifications development, market analysis, productivity/cost...
-
A digital Video Camera provides an uncompressed output provide a signal resolution of 720 X 480 pixels at a frame rate of 30 fps and 16-bit quantization for each pixel What is the required bandwidth...
-
d. The characteristic equation of a control system is given by s+2s+8s+12s+20s+16+16=0. Determine the number of the roots of the equation which lie on the imaginary axis of s-plane
-
1. Why has there been a corporate governance "revolution" throughout the world recently? 2. What are the main areas of deficiencies as pointed out by corporate governance reformers? 3. Representing...
-
After watching the video on Cubas economy, discuss the following: 1. Can Cuba effectively become a free market economy? 2. Could the US benefit from lifting the embargo on Cuba? 3. What are Cubas...
-
After watching the video on interest rates at the European Central Bank, discuss the following: 1. How will the rise in interest rates affect FDI for these euro zone countries? 2. What reasons can...
-
Give an example of a local wind pattern. Give an example of a global wind pattern.
-
What drives surface currents?
-
Do surface currents affect water temperature, air temperature, or both?

Study smarter with the SolutionInn App


