Ascertain, with a little calculation, whether or not each of the following reactions will proceed to...
Fantastic news! We've Found the answer you've been seeking!
Question:
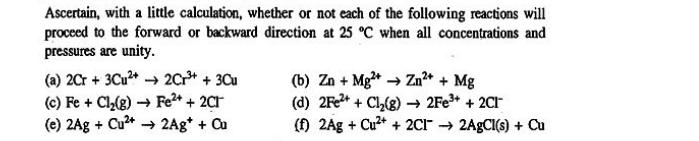
Transcribed Image Text:
Ascertain, with a little calculation, whether or not each of the following reactions will proceed to the forward or backward direction at 25 °C when all concentrations and pressures are unity. (a) 2Cr + 3Cu²+→→2Cr³+ + 3Cu (c) Fe + Cl₂(g) →→ Fe²+ + 2Cl (e) 2Ag + Cu²+ 2Ag* + Cu (b) Zn + Mg²+→→ Zn²+ + Mg (d) 2Fe²+ + Cl₂(g) →→2Fe³+ + 2Cl (f) 2Ag + Cu²+ + 2C12AgCl(s) + Cu Ascertain, with a little calculation, whether or not each of the following reactions will proceed to the forward or backward direction at 25 °C when all concentrations and pressures are unity. (a) 2Cr + 3Cu²+→→2Cr³+ + 3Cu (c) Fe + Cl₂(g) →→ Fe²+ + 2Cl (e) 2Ag + Cu²+ 2Ag* + Cu (b) Zn + Mg²+→→ Zn²+ + Mg (d) 2Fe²+ + Cl₂(g) →→2Fe³+ + 2Cl (f) 2Ag + Cu²+ + 2C12AgCl(s) + Cu
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Each of the following reactions will be encountered at some point in this text. Classify each one according to whether the organic substrate is oxidized or reduced in the process. (a) CH3C = PCH +...
-
Determine whether or not each of the following signals is periodic. In case a signal is periodic, specify its fundamental period. (a) xa(t) = 3 cos(5t + /6) (b) x(n) = 3 cos(5n + /6) (c) x(n) = 2...
-
Indicate whether or not each of the following ten items should be included in the cash balance presented on the balance sheet. Also indicate the normal balance sheet treatment for those items not...
-
Why is it important to have a defined project scope? Why is it important to make sure there is agreement about the scope? Is there anything in the "Why Should You Use the WBS?
-
Explain the following statement: Horizontal consistency of goals is as important as vertical consistency.
-
An overflow spillway with a 100-m-long crest will convey a design discharge of 1,150 m3/sec under a permitted maximum head of 3.00 m. The operation of the prototype spillway is studied on a 1:50...
-
Atlanta Corp. is preparing its 2020 financial statements (December 31 year-end) and is considering the following events: a. Probable warranty costs on the company's products are estimated to be \(1...
-
Tombert Decorating uses a job order cost system to collect the costs of its interior decorating business. Each clients consultation is treated as a separate job. Overhead is applied to each job based...
-
4 Common-size and trend percentages for a company's net sales, cost of goods sold, and expenses follow: Common-Size Percentages Trend Percentages Net sales 2023 100.0% 2022 100.0% 2021 2023 20 Cost...
-
On December 1, a six-month liability insurance policy was purchased for $900. Analyze the required adjustment as of December 31 using T accounts, and then formally enter this adjustment in the...
-
(3) Stupid Valley Bank (SVB) has reserves of $1,000, deposits of $22,000, debts of $19,000, loans of $21,000, and capital (owner's equity) of $1,000. In addition, the bank purchased a 52 weeks (364...
-
The contour diagram of a function z=f(x,y) is given below. Determine the sign of the second partial derivatives at the point P. Note the contours below all have negative values. 3.5 3 2.5 -8- 9 ---8-...
-
A 33.0 kg beam is attached to a wall with a hinge while its far end is supported by a cable such that the beam is horizontal. If the angle between the beam and the cable is = 55.0 what is the...
-
Required Information [The following Information applies to the questions displayed below.] As of December 31 of the current year, Armani Company's records show the following. Hint. The owner Invested...
-
Arthur Moore commenced business on 1 October by paying 50,000 into a newly opened business bank account. His transactions during the first three weeks of business were as follows; Purchased fixtures...
-
Using the IST Linux system create the following Java command line application in your home area Lab7.java. Accept user input from the command line and check for an integer type and handle the...
-
Inanother- much larger - study,a 10 g/gincrease in tibialead was associatedwitha 0.55mm Hg increase in systolicBPin girlsafteradjustingfora set ofpotentiallyrelevantconfounders.Assume that 0.55is...
-
Activator rod AB exerts on crank BCD a force P directed along line AB. Knowing that P must have a 100-N component perpendicular to arm BC of the crank, determine (a) The magnitude of the force P, (b)...
-
Î, γUnsaturated steroidal ketones represented by the partial structure shown here are readily converted in acid to their α, β -unsaturated isomers. Write a...
-
Poly(vinyl alcohol) is a useful water-soluble polymer. It cannot be prepared directly from vinyl alcohol, because of the rapidity with which vinyl alcohol (CH2==CHOH) isomerizes to acetaldehyde....
-
In the simplest molecular orbital treatment of conjugated systems, it is assumed that the Ï system does not interact with the framework of Ï bonds. When this MO method was used to calculate...
-
6. One afternoon while visiting friends, tennis star Vitas Gerulaitis fell asleep in their pool house. A mechanic had improperly installed the swimming pool heater, which leaked carbon monoxide fumes...
-
5. One Friday afternoon a custodian at the Lazear Elementary School in Oakland, California, raped an 11-year-old student in his office on the school premises. The
-
8. A. B. Rains worked as a broker for the Joseph Denunzio Fruit Co. Raymond Crane offered to sell Rains nine carloads of emperor grapes. Rains accepted the offer on behalf of Denunzio. Later, Rains...

Study smarter with the SolutionInn App



