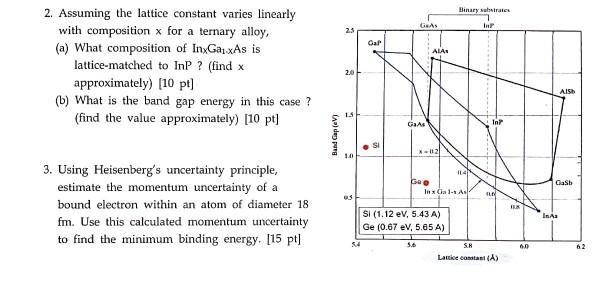
2. Assuming the lattice constant varies linearly with composition x for a ternary alloy, (a) What...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
2. Assuming the lattice constant varies linearly with composition x for a ternary alloy, (a) What composition of InxGaxAs is lattice-matched to InP ? (find x approximately) [10 pt] (b) What is the band gap energy in this case ? (find the value approximately) [10 pt] 3. Using Heisenberg's uncertainty principle, estimate the momentum uncertainty of a bound electron within an atom of diameter 18 fm. Use this calculated momentum uncertainty to find the minimum binding energy. [15 pt] Band Gap (EV) 23 2.0 15 1.0 05 Gal ● SI GRAS GaAs AIA *-02 Goo Binary substrates Si (1.12 eV, 5.43 A) Ge (0.67 eV, 5.65 A) 114 Inx Ga 1-1 As InP In FLO 5.8 Lattice constant (A) BUK 60 AISH Gasb Ins 62 2. Assuming the lattice constant varies linearly with composition x for a ternary alloy, (a) What composition of InxGaxAs is lattice-matched to InP ? (find x approximately) [10 pt] (b) What is the band gap energy in this case ? (find the value approximately) [10 pt] 3. Using Heisenberg's uncertainty principle, estimate the momentum uncertainty of a bound electron within an atom of diameter 18 fm. Use this calculated momentum uncertainty to find the minimum binding energy. [15 pt] Band Gap (EV) 23 2.0 15 1.0 05 Gal ● SI GRAS GaAs AIA *-02 Goo Binary substrates Si (1.12 eV, 5.43 A) Ge (0.67 eV, 5.65 A) 114 Inx Ga 1-1 As InP In FLO 5.8 Lattice constant (A) BUK 60 AISH Gasb Ins 62
Expert Answer:

Related Book For 

Essentials of Materials Science and Engineering
ISBN: 978-1111576851
3rd edition
Authors: Donald R. Askeland, Wendelin J. Wright
Posted Date:
Students also viewed these electrical engineering questions
-
A potassium chloride crystal has an energy band gap of 7.6 eV above the topmost occupied band, which is full. Is this crystal opaque or transparent to light of wavelength 140 nm?
-
What is comprehensive income? Its composition varies from company to company but may include which investment-related items that are not included in net income?
-
A copper-nickel alloy of composition 70 wt% Ni-30 wt% Cu is slowly heated from a temperature of 1300C (2370F). (a) At what temperature does the first liquid phase form? (b) What is the composition of...
-
Using the 526 observations on workers in a certain dataset, we include educ (years of education), exper (years of labor market experience) and tenure (years with the current employer) in regression...
-
Explain why investors who have diversified their portfolios will determine the price and, consequently, the expected return on an asset.
-
Derek dies intestate (i.e., without a will) and is survived by a daughter, Ruth, and a grandson, Ted (Ruth's son). Derek's assets include a large portfolio of stocks and bonds and a beach house. Ruth...
-
T Morgan, a sole trader, extracted the following trial balance from his books at the close of business on 31 March 19X9: Notes: a) Stock 31 March 19X9 4,290. b) Wages and salaries accrued at 31 March...
-
A U.S. Postal Service supervisor is looking for ways to reduce stress in the sorting department. With the existing arrangement stamped letters are machine-canceled and loaded into tubs with 375...
-
In the Better Business Bureau settled ofcomplaints they received in the United States. Suppose you havebeen hired by the Better Business Bureau to investigate thecomplaints they received this yea 2...
-
The equilibrium adsorption of methane on a given activated carbon was studied by Grant et al. (1962). They proposed a Langmuir-type adsorption isotherm with parameters \(q_{m}=48 \mathrm{~g}...
-
Samantha's Office Supplies manufactures desk organizers in its Processing Department. Direct materials are included at the inception of the production cycle and must be bundled in single kits for...
-
38. In the given diagram meters are not ideal. If reading of ammeter and voltmeter are 6A and 30V respectively then :- ww-A R (1) R=50 (2) R>50 (3) R <50 (4) All the above are possible
-
Assume you wrote a report on employee education and training in your company. Write an executive summary about your report on employee education and training in your company.
-
Provide an example of a completed post trail validation tool for TAE 40116 Assessment cluster.
-
what variable must you alter on the graph to change the evaporation rate? explain in detail why this variable influences evaporation rate
-
. A sample of hydrogen fluoride is placed in a 1.00 L container. At equilibrium, the concentration of HF (g) was 0.700 mol/L. The equilibrium constant for the formation of hydrogen fluoride gas is...
-
1. After reading what was most memorable and what did you learn? Leticia Renteria is a 30-year-old woman whoalong with her husband, Marcos Vargasis a parent to three young children, ages 4, 5, and 7....
-
The graph of the sequence whose general term is an = n - 1 is which of the following? [8.1] A. B. TITTT 3-2-1 23.45 2.3.4
-
A polyimide matrix with an elastic modulus of 0.3 106 psi is to be reinforced with 70 vol% carbon fibers to give a minimum modulus of elasticity of 40 106 psi. Recommend a process for producing the...
-
A 1.2% C tool steel held at 1150 C is exposed to oxygen for 48 h. The carbon content at the steel surface is zero. To what depth will the steel be decarburized to less than 0.20% C?
-
The following data were collected from a 20-mm-dia meter test specimen of a ductile cast iron (l0 = 40.00 mm): After fracture, the total length was 47.42 mm , and the diameter was 18.35 mm. Plot the...
-
In a survey of U.S. college students, 74% believed that colleges do not have the right to restrict the expression of political views on campus. You randomly select 20 college students and ask them if...
-
In a sample of 100 eruptions of the Old Faithful geyser at Yellowstone National Park, the mean interval between eruptions was 129.58 minutes and the standard deviation was 108.54 minutes. A random...
-
Twenty-two percent of adults over 18 years of age have a bachelors degree. You randomly select 20 adults over 18 years of age and ask whether they have a bachelors degree. In Exercises 17 and 18, a...

Study smarter with the SolutionInn App


