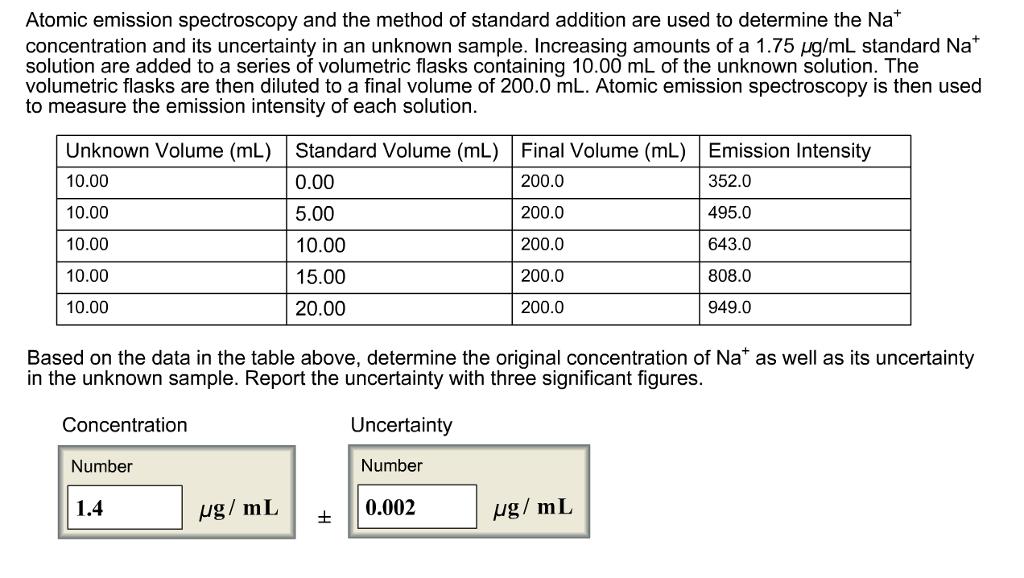
Atomic emission spectroscopy and the method of standard addition are used to determine the Na* concentration...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Atomic emission spectroscopy and the method of standard addition are used to determine the Na* concentration and its uncertainty in an unknown sample. Increasing amounts of a 1.75 ug/mL standard Na* solution are added to a series of volumetric flasks containing 10.00 mL of the unknown solution. The volumetric flasks are then diluted to a final volume of 200.0 mL. Atomic emission spectroscopy is then used to measure the emission intensity of each solution. Unknown Volume (mL) Standard Volume (mL) Final Volume (mL) Emission Intensity 10.00 0.00 200.0 352.0 10.00 5.00 200.0 495.0 10.00 10.00 200.0 643.0 10.00 15.00 200.0 808.0 10.00 20.00 200.0 949.0 Based on the data in the table above, determine the original concentration of Na* as well as its uncertainty in the unknown sample. Report the uncertainty with three significant figures. Concentration Uncertainty Number Number 1.4 µg/ mL 0.002 ug/ mL Atomic emission spectroscopy and the method of standard addition are used to determine the Na* concentration and its uncertainty in an unknown sample. Increasing amounts of a 1.75 ug/mL standard Na* solution are added to a series of volumetric flasks containing 10.00 mL of the unknown solution. The volumetric flasks are then diluted to a final volume of 200.0 mL. Atomic emission spectroscopy is then used to measure the emission intensity of each solution. Unknown Volume (mL) Standard Volume (mL) Final Volume (mL) Emission Intensity 10.00 0.00 200.0 352.0 10.00 5.00 200.0 495.0 10.00 10.00 200.0 643.0 10.00 15.00 200.0 808.0 10.00 20.00 200.0 949.0 Based on the data in the table above, determine the original concentration of Na* as well as its uncertainty in the unknown sample. Report the uncertainty with three significant figures. Concentration Uncertainty Number Number 1.4 µg/ mL 0.002 ug/ mL
Expert Answer:
Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog,Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date:
Students also viewed these chemistry questions
-
The method of standard additions was used to determine nitrite in a soil sample. A 1.00-mL portion of the sample was mixed with 24.00 mL of a colorimetric reagent, and the nitrite was converted to a...
-
In Monopoly, dice are used to determine which player gets to move first. Suppose there are two players in the game. Each player rolls a die and the player with the higher number gets to move first....
-
An analyst plans to use P/E and the method of comparables as a basis for recommending purchasing shares of one of two peer - group companies in the business of manufacturing personal digital...
-
In 2020, Neighbor Co-Op Inc. sells 1,100 beverages in glass bottles and receives a $1.00 deposit for each returnable bottle sold. As of December 31, 2020, a total of 880 glass bottles were returned...
-
For each of the following statements, indicate whether it is true or false. a. A spontaneous reaction always releases heat. b. A spontaneous reaction is always a fast reaction. c. The entropy of a...
-
Sunnyfield Industries will lease a new rock crusher for $30,000 per year for 5 years, paid at the end of each year. These lease terms are consistent with an annual interest rate of 6%. How will the...
-
If an IEC 61508 certified logic solver is selected, what prior use safety integrity justification is required?
-
Discuss whether the changes described in each of the cases below require recognition in the CPAs report as to consistency. (Assume that the amounts are material.) (a) After 3 years of computing...
-
Moran owns a building he bought during year 0 for $150,000. He sold the building in year 6. During the time he held the building, he depreciated it by $32,000. What are the amount and character of...
-
Batch processes are often used in chemical and pharmaceutical operations to achieve a desired chemical composition for the final product. Related heat transfer processes are typically transient,...
-
A patient and her spouse have been attempting to maintain a pregnancy for more than 20 weeks for the past 12 months. Her spouse has had a sperm analysis performed and it was WNL The patient can jet...
-
Review the Peloton situation and email in Figure 3. Imagine that youre an executive coach for the CEO of Peloton. Describe the steps in the writing process that he should take to ensure that future...
-
Throughout the day, keep a list of emotions you see on peoples faces. Try to be more precise than happy, sad, or angry. People might feel upset, delighted, anxious, determined, grateful, ambitious,...
-
If your country of origin is different from where youre studying, you may volunteer to describe the culture to the class. Or you may volunteer if you have traveled to another country. Answer the...
-
Prepare to write a review of a product you used or purchased recently. Use the process outlined in this chapter: Brainstorm ideas. What do you think is important to include in your review? Draw a...
-
Next time youre on a video meeting, record yourself speaking and listening. Watch yourself later to observe your nonverbal communication. Are your words aligned with your facial expressions,...
-
Discuss several different types of media that could and would be beneficial to law enforcement and why you think so. Discuss the complexity of policemedia relations, especially as this relates to...
-
Solve each equation. x 3 - 6x 2 = -8x
-
Why is a Walden reductor always used with solutions that contain appreciable concentrations of HCl?
-
List the types of substances to which each of the following separation methods is most applicable: (a) Supercritical fluid chromatography. (b) Capillary zone electrophoresis. (c) Flow FFF.
-
Consider curves for the titration of 0.10 M NaOH and 0.010 M NH3 with 0.10 M HCl. (a) Briefly account for the differences between curves for the two titrations. (b) In what respect will the two...
-
Consider an experiment that selects a cell phone camera and records the recycle time of a flash (the time taken to ready the camera for another flash). The possible values for this time depend on the...
-
Suppose that the recycle times of two cameras are recorded. The extension of the positive real line \(R\) is to take the sample space to be the positive quadrant of the plane \[ S=R^{+} \times R^{+}...
-
Consider the sample space \(S=\{y y, y n, n y, n n\}\) in Example 2.2. Suppose that the subset of outcomes for which at least one camera conforms is denoted as \(E_{1}\). Then, \[ E_{1}=\{y y, y n, n...

Study smarter with the SolutionInn App



