b) A system consists of air initially at state 1, where P = 100 kPa, T...
Fantastic news! We've Found the answer you've been seeking!
Question:

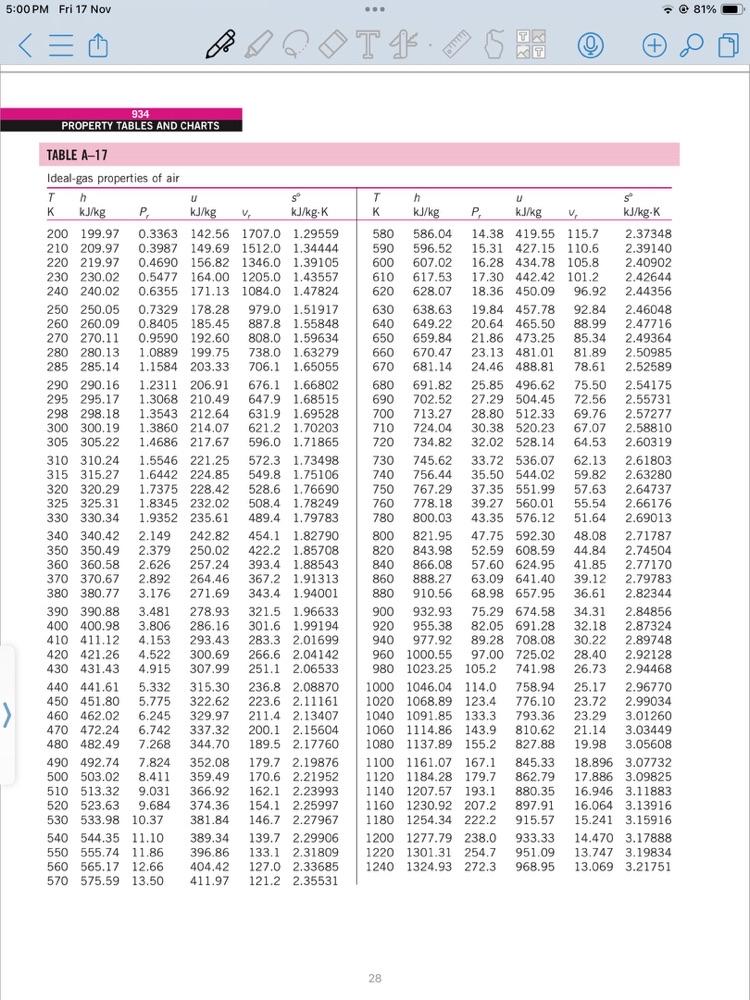
Transcribed Image Text:
b) A system consists of air initially at state 1, where P = 100 kPa, T = 350K. The system undergoes the following process: Process 1 Process 2 2: adiabatic compression to P2 = 1500 kPa, v = 0.1359 m/kg 3: constant volume cooling to T3 = 300K a) Show P-v diagram b) Determine work and heat transfer for each process using data from Table A-17 and constant specific heat c) Discuss your findings (10 marks) 5:00 PM Fri 17 Nov 934 PROPERTY TABLES AND CHARTS TABLE A-17 Ideal-gas properties of air P T - 81% (+ Th U T h U K kJ/kg P kJ/kg V kJ/kg-K K kJ/kg P kJ/kg V kJ/kg-K 200 199.97 0.3363 142.56 1707.0 1.29559 580 586.04. 14.38 419.55 115.7 2.37348 210 209.97 0.3987 149.69 1512.0 1.34444 220 219.97 0.4690 156.82 1346.0 1.39105 590 596.52 15.31 427.15 110.6 2.39140 600 607.02 16.28 434.78 105.8 2.40902 230 230.02 0.5477 164.00 1205.0 1.43557 610 617.53 17.30 442.42 101.2 2.42644 240 240.02 0.6355 171.13 1084.0 1.47824 620 628.07 18.36 450.09 96.92 2.44356 250 250.05 0.7329 178.28 979.0 1.51917 630 638.63 19.84 457.78 92.84 2.46048 260 260.09 0.8405 185.45 270 270.11 0.9590 192.60 280 280.13 1.0889 199.75 285 285.14 1.1584 203.33 290 290.16 1.2311 206.91 295 295.17 1.3068 210.49 647.9 1.68515 298 298.18 1.3543 212.64 631.9 1.69528 300 300.19 1.3860 214.07 621.2 1.70203 305 305.22 1.4686 217.67 596.0 1.71865 310 310.24 1.5546 221.25 572.3 1.73498 315 315.27 1.6442 224.85 549.8 1.75106 320 320.29 1.7375 228.42 528.6 1.76690 325 325.31 1.8345 232.02 508.4 1.78249 330 330.34 1.9352 235.61 489.4 1.79783 340 340.42 2.149 242.82 454.1 1.82790 350 350.49 2.379 250.02 422.2 1.85708 360 360.58 2.626 257.24 393.4 1.88543 370 370.67 2.892 264.46 367.2 1.91313. 380 380.77 3.176 271.69 343.4 1.94001 390 390.88 3.481 278.93 321.5 1.96633 400 400.98 3.806 286.16 301.6 1.99194 410 411.12 4.153 293.43 283.3 2.01699 420 421.26 4.522 300.69 266.6 2.04142 430 431.43 4.915 307.99 251.1 2.06533 440 441.61 5.332 315.30 236.8 2.08870 450 451.80 5.775 322.62 223.6 2.11161 460 462.02 6.245 329.97 211.4 2.13407 470 472.24 6.742 337.32 200.1 2.15604 480 482.49 7.268 344.70 189.5 2.17760 490 492.74 7.824 352.08 179.7 2.19876 500 503.02 8.411 359.49 170.6 2.21952 510 513.32 9.031 366.92 162.1 2.23993 520 523.63 9.684 374.36 154.1 2.25997 530 533.98 10.37 381.84 146.7 2.27967 540 544.35 11.10 550 555.74 11.86 560 565.17 12.66 570 575.59 13.50 887.8 1.55848 640 649.22 20.64 465.50 88.99 2.47716 808.0 1.59634 650 659.84. 21.86 473.25 85.34 2.49364 738.0 1.63279 706.1 1.65055 660 670.47 23.13 481.01 81.89 2.50985 670 681.14 24.46 488.81 78.61 2.52589 676.1 1.66802 680 691.82 25.85 496.62 75.50 2.54175 690 702.52 27.29 504.45 72.56 2.55731 700 713.27 28.80 512.33 69.76 2.57277 710 724.04 30.38 520.23 67.07 2.58810 720 730 740 756.44 750 767.29 760 778.18 780 800.03 800 821.95 734.82 32.02 528.14 64.53 2.60319 745.62 33.72 536.07 62.13 2.61803 35.50 544.02 37.35 551.99. 57.63 39.27 560.01 43.35 576.12 59.82 2.63280 2.64737 55.54 2.66176 51.64 2.69013 47.75 592.30 48.08 2.71787 820 843.98 52.59 608.59 44.84 2.74504 840 860 866.08 57.60 624.95 41.85 2.77170 888.27 63.09 641.40 39.12 2.79783 880 910.56 900 920 68.98 657.95 36.61 2.82344 932.93 955.38 75.29 674.58 34.31 2.84856 82.05 691.28 32.18 2.87324 940 977.92 389.34 396.86 139.7 2.29906 133.1 2.31809 404.42 411.97 127.0 2.33685 121.2 2.35531 960 1000.55 980 1023.25 105.2 1000 1046.04 114.0 1020 1068.89 123.4 1040 1091.85 133.3 1060 1114.86 143.9 1080 1137.89 155.2 1100 1161.07 167.1 1120 1184.28 179.7 1140 1207.57 193.1 1160 1230.92 207.2 1180 1254.34 222.2 1200 1277.79 238.0 1220 1301.31 254.7 1240 1324.93 272.3 89.28 708.08 97.00 725.02 741.98 758.94 25.17 2.96770 776.10 23.72 2.99034 793.36 23.29 3.01260 810.62 21.14 3.03449 827.88 19.98 3.05608 845.33 18.896 3.07732 862.79 880.35 897.91 30.22 2.89748 28.40 2.92128 26.73 2.94468 17.886 3.09825 16.946 3.11883 16.064 3.13916 915.57 15.241 3.15916 933.33 14.470 3.17888 951.09 968.95 13.747 3.19834 13.069 3.21751 28 b) A system consists of air initially at state 1, where P = 100 kPa, T = 350K. The system undergoes the following process: Process 1 Process 2 2: adiabatic compression to P2 = 1500 kPa, v = 0.1359 m/kg 3: constant volume cooling to T3 = 300K a) Show P-v diagram b) Determine work and heat transfer for each process using data from Table A-17 and constant specific heat c) Discuss your findings (10 marks) 5:00 PM Fri 17 Nov 934 PROPERTY TABLES AND CHARTS TABLE A-17 Ideal-gas properties of air P T - 81% (+ Th U T h U K kJ/kg P kJ/kg V kJ/kg-K K kJ/kg P kJ/kg V kJ/kg-K 200 199.97 0.3363 142.56 1707.0 1.29559 580 586.04. 14.38 419.55 115.7 2.37348 210 209.97 0.3987 149.69 1512.0 1.34444 220 219.97 0.4690 156.82 1346.0 1.39105 590 596.52 15.31 427.15 110.6 2.39140 600 607.02 16.28 434.78 105.8 2.40902 230 230.02 0.5477 164.00 1205.0 1.43557 610 617.53 17.30 442.42 101.2 2.42644 240 240.02 0.6355 171.13 1084.0 1.47824 620 628.07 18.36 450.09 96.92 2.44356 250 250.05 0.7329 178.28 979.0 1.51917 630 638.63 19.84 457.78 92.84 2.46048 260 260.09 0.8405 185.45 270 270.11 0.9590 192.60 280 280.13 1.0889 199.75 285 285.14 1.1584 203.33 290 290.16 1.2311 206.91 295 295.17 1.3068 210.49 647.9 1.68515 298 298.18 1.3543 212.64 631.9 1.69528 300 300.19 1.3860 214.07 621.2 1.70203 305 305.22 1.4686 217.67 596.0 1.71865 310 310.24 1.5546 221.25 572.3 1.73498 315 315.27 1.6442 224.85 549.8 1.75106 320 320.29 1.7375 228.42 528.6 1.76690 325 325.31 1.8345 232.02 508.4 1.78249 330 330.34 1.9352 235.61 489.4 1.79783 340 340.42 2.149 242.82 454.1 1.82790 350 350.49 2.379 250.02 422.2 1.85708 360 360.58 2.626 257.24 393.4 1.88543 370 370.67 2.892 264.46 367.2 1.91313. 380 380.77 3.176 271.69 343.4 1.94001 390 390.88 3.481 278.93 321.5 1.96633 400 400.98 3.806 286.16 301.6 1.99194 410 411.12 4.153 293.43 283.3 2.01699 420 421.26 4.522 300.69 266.6 2.04142 430 431.43 4.915 307.99 251.1 2.06533 440 441.61 5.332 315.30 236.8 2.08870 450 451.80 5.775 322.62 223.6 2.11161 460 462.02 6.245 329.97 211.4 2.13407 470 472.24 6.742 337.32 200.1 2.15604 480 482.49 7.268 344.70 189.5 2.17760 490 492.74 7.824 352.08 179.7 2.19876 500 503.02 8.411 359.49 170.6 2.21952 510 513.32 9.031 366.92 162.1 2.23993 520 523.63 9.684 374.36 154.1 2.25997 530 533.98 10.37 381.84 146.7 2.27967 540 544.35 11.10 550 555.74 11.86 560 565.17 12.66 570 575.59 13.50 887.8 1.55848 640 649.22 20.64 465.50 88.99 2.47716 808.0 1.59634 650 659.84. 21.86 473.25 85.34 2.49364 738.0 1.63279 706.1 1.65055 660 670.47 23.13 481.01 81.89 2.50985 670 681.14 24.46 488.81 78.61 2.52589 676.1 1.66802 680 691.82 25.85 496.62 75.50 2.54175 690 702.52 27.29 504.45 72.56 2.55731 700 713.27 28.80 512.33 69.76 2.57277 710 724.04 30.38 520.23 67.07 2.58810 720 730 740 756.44 750 767.29 760 778.18 780 800.03 800 821.95 734.82 32.02 528.14 64.53 2.60319 745.62 33.72 536.07 62.13 2.61803 35.50 544.02 37.35 551.99. 57.63 39.27 560.01 43.35 576.12 59.82 2.63280 2.64737 55.54 2.66176 51.64 2.69013 47.75 592.30 48.08 2.71787 820 843.98 52.59 608.59 44.84 2.74504 840 860 866.08 57.60 624.95 41.85 2.77170 888.27 63.09 641.40 39.12 2.79783 880 910.56 900 920 68.98 657.95 36.61 2.82344 932.93 955.38 75.29 674.58 34.31 2.84856 82.05 691.28 32.18 2.87324 940 977.92 389.34 396.86 139.7 2.29906 133.1 2.31809 404.42 411.97 127.0 2.33685 121.2 2.35531 960 1000.55 980 1023.25 105.2 1000 1046.04 114.0 1020 1068.89 123.4 1040 1091.85 133.3 1060 1114.86 143.9 1080 1137.89 155.2 1100 1161.07 167.1 1120 1184.28 179.7 1140 1207.57 193.1 1160 1230.92 207.2 1180 1254.34 222.2 1200 1277.79 238.0 1220 1301.31 254.7 1240 1324.93 272.3 89.28 708.08 97.00 725.02 741.98 758.94 25.17 2.96770 776.10 23.72 2.99034 793.36 23.29 3.01260 810.62 21.14 3.03449 827.88 19.98 3.05608 845.33 18.896 3.07732 862.79 880.35 897.91 30.22 2.89748 28.40 2.92128 26.73 2.94468 17.886 3.09825 16.946 3.11883 16.064 3.13916 915.57 15.241 3.15916 933.33 14.470 3.17888 951.09 968.95 13.747 3.19834 13.069 3.21751 28
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
(iii) Now express your decision rule instead using only the quantities p(Ck), p(Cj ), p(x|Ck), p(x|Cj ), and relate it to the diagram above. [2 marks] (iv) If the classifier decision rule assigns...
-
What is GST?
-
Question 6.5 reported the following regression (where standard errors have been added): a. Is the coefficient on BDR statistically significantly different from zero? b. Typically five-bedroom houses...
-
Does the current Social Security system promote income equality? Why or why not?
-
(a) Imagine holding a ball a certain height above the ground. If you let the ball go, it accelerates downward. An interaction between the ball and what other object causes this acceleration? Is this...
-
The transactions that follow took place at the Cedar Hill Sports Arena during September 2016. This firm has indoor courts where customers can play tennis for a fee. It also rents equipment and offers...
-
6. An igloo, a hemispherical enclosure built of ice (1.67 J/m-sC), has as inner radius of 2.50m. The thickness of the ice is 0.5m. At what rate must thermal energy be generated to maintain the air...
-
How much will you charge for your product or service? What are the major costs of doing this business? Include any assumptions you use in generating cost estimates. How much money will you need to...
-
Find an organizational chart for your employer or a company that you are familiar with. Is it primarily functional or process-focused? Explain.
-
Name Due Date Assignment Solutions to the quadratic equation Ax +Bx + C = 0 may be calculated using the quadratic formulas: x = -B+B2-4AC 2A -B-B-4AC 2A These formulas may be used, of course, only if...
-
Explain positional bargaining strategy between management and union on multi-shift operations in manufacturing companies
-
What are the benefits of volunteerism for older adult cognitive, physical and social well-being. Discuss.
-
What is the responsibility of the healthcare provider to identify and address the risk factors for social isolation within older adults? Explain.
-
Explain the importance and principal responsibilities of audit committees with particular reference to auditors' independence.
-
For all of the following words, if you move the first letter to the end of the word, and then spell the result backwards, you will get the original word: banana dresser grammar potato revive uneven...
-
Verify that \(\left|\Delta v_{\mathrm{u} x} ight| /\left|\Delta v_{\mathrm{s} x} ight| \approx 1 / 3\) for the two carts in Figure 4.9. Figure 4.9 Velocity-versus-time graph for a standard cart and a...
-
What is the ratio of the \(x\) components of the change in velocity for the plastic and metal carts, \(\Delta v_{\mathrm{p} x} / \Delta v_{\mathrm{m} x}\), in Figure 4.10? Figure 4.10...
-
Is the inertia of the cart of unknown inertia in Figure 4.9 greater or less than that of the standard cart? Figure 4.9 Velocity-versus-time graph for a standard cart and a cart of unknown inertia...

Study smarter with the SolutionInn App


