Question: B. Enzo, Hotel manager, would like to develop a cost model to predict the future costs of running the hotel. Unfortunately, the only available
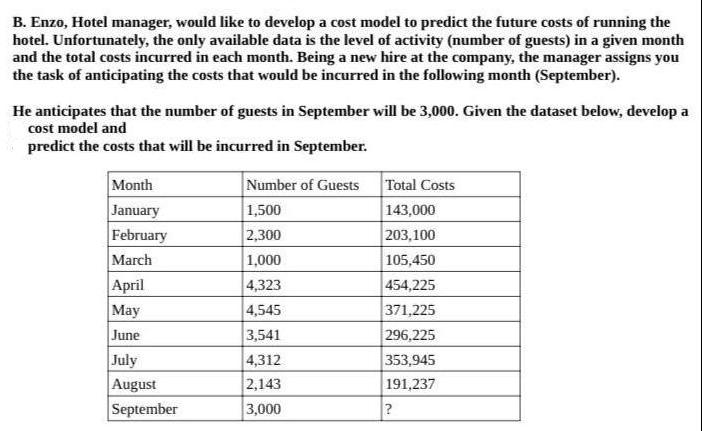
B. Enzo, Hotel manager, would like to develop a cost model to predict the future costs of running the hotel. Unfortunately, the only available data is the level of activity (number of guests) in a given month and the total costs incurred in each month. Being a new hire at the company, the manager assigns you the task of anticipating the costs that would be incurred in the following month (September). He anticipates that the number of guests in September will be 3,000. Given the dataset below, develop a cost model and predict the costs that will be incurred in September. Month January February March April May June July August September Number of Guests 1,500 2,300 1,000 4,323 4,545 3,541 4,312 2,143 3,000 Total Costs 143,000 203,100 105,450 454,225 371,225 296,225 353,945 191,237 ?
Step by Step Solution
3.51 Rating (151 Votes )
There are 3 Steps involved in it
To develop a cost model we can use the highlow method to estimate the fixed and variable components ... View full answer

Get step-by-step solutions from verified subject matter experts


