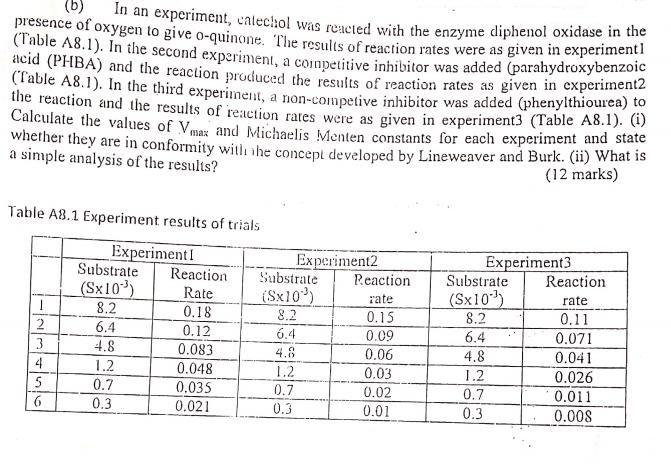
(b) presence of oxygen to give o-quinone. The results of reaction rates were as given in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
(b) presence of oxygen to give o-quinone. The results of reaction rates were as given in experimenti (Table A8.1). In the second experiment, a coupetitive inhibitor was added (parahydroxybenzoe acid (PHBA) and the reaction produced the results of reaction rates as given in experimentz (Table A8.1). In the third experiment, a non-competive inhibitor was added (phenylthioure) the reaction and the results of reaction rates were as given in experiment3 (Table A8.1). (1) Calculate the values of Vmaz and Michaelis Menten constants for each experiment and state wherher they are in conformity witi he concept developed by Lineweaver and Burk. (ii) What is a simple analysis of the results? In an experiment, calechol was reacted with the enzyme diphenol oxidase in the (12 marks) Table A8.1 Experiment results of trials ExperimentI Reaction Rate 0.18 0.12 Experiment2 Substrate Experiment3 Substrate Substrate Reaction Reaction (Sx10) 8,2 (Sx10*) (Sx10) 8.2 rate rate 8.2 0.15 0.11 6.4 6.4 0.09 6.4 0.071 3 4.8 0.083 0.06 0.041 0.026 4.8 4.8 4 1.2 0.048 1.2 0.03 1.2 5 0.7 0.035 0.7 0.02 0.7 0.011 6. 0.3 0.021 0.3 0.01 0.3 0.008 (b) presence of oxygen to give o-quinone. The results of reaction rates were as given in experimenti (Table A8.1). In the second experiment, a coupetitive inhibitor was added (parahydroxybenzoe acid (PHBA) and the reaction produced the results of reaction rates as given in experimentz (Table A8.1). In the third experiment, a non-competive inhibitor was added (phenylthioure) the reaction and the results of reaction rates were as given in experiment3 (Table A8.1). (1) Calculate the values of Vmaz and Michaelis Menten constants for each experiment and state wherher they are in conformity witi he concept developed by Lineweaver and Burk. (ii) What is a simple analysis of the results? In an experiment, calechol was reacted with the enzyme diphenol oxidase in the (12 marks) Table A8.1 Experiment results of trials ExperimentI Reaction Rate 0.18 0.12 Experiment2 Substrate Experiment3 Substrate Substrate Reaction Reaction (Sx10) 8,2 (Sx10*) (Sx10) 8.2 rate rate 8.2 0.15 0.11 6.4 6.4 0.09 6.4 0.071 3 4.8 0.083 0.06 0.041 0.026 4.8 4.8 4 1.2 0.048 1.2 0.03 1.2 5 0.7 0.035 0.7 0.02 0.7 0.011 6. 0.3 0.021 0.3 0.01 0.3 0.008
Expert Answer:
Answer rating: 100% (QA)
From the graph Vmax 01272 in units of rate and KM 0001585 in units of substrate conc Link to ... View the full answer

Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog,Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date:
Students also viewed these chemical engineering questions
-
An enzyme contains an active site aspartic acid with a pKa = 5.0, which acts as a general acid catalyst. On the template below, draw the curve of enzyme activity (reaction rate) versus pH for the...
-
Rate constants for a reaction were determined at five temperatures. From the following data, calculate the experimental energy of activation and then calculate G¡, H¡, and S¡ for...
-
A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons. (a) Draw the resonance forms of a carboxylic acid that is protonated on the hydroxyl oxygen atom. (b) Compare the...
-
The Globe and Mail compiled a list of the top 100 public companies in Canada according to profit. Leading the list is the Toronto- Dominion Bank, followed by the Bank of Nova Scotia. The following...
-
Use each of the Adams-Bashforth methods to approximate the solutions to the following initial-value problems. In each case use starting values obtained from the Runge-Kutta method of order four....
-
Free doesn't always win. Why might a firm turn down free software in favor of a commercial alternative?
-
It is desired to have \($9,000\) available 12 yr from now. If \($5,000\) is available for investment at the present time, what discrete annual rate of compound interest on the investment would be...
-
Gemini, Inc., an all-equity firm, is considering a $1.7 million investment that will be depreciated according to the straight-line method over its four-year life. The project is expected to generate...
-
The expected dividend per share on the equity share of Roadport Limited is $2.00. The dividend per share of Roadport Limited has grown over the past five years at the rate of 5% per year. This growth...
-
A thin smooth disk of diameter D is immersed parallel to a uniform stream of velocity U. Assuming laminar flow and using flat-plate theory as a guide, develop an approximate formula for the drag of...
-
Create journal entries for all other (non-inventory) owner contributions View the Video: Owner Contributions / Investments To enter all owner contributions, you may create one or multiple JEs....
-
To reduce an anticipated large number of applicants for a graphic designer position, a senior recruiter adds proficiency in the graphics design software she remembers learning about when she was in...
-
Discuss and analyze the effectiveness of the "The Barbershop Girls of India" campaign using the 6M framework for marketing communications, with citations?
-
As a supply chain student, briefly explain how you think the technology has impacted supply chain in the last decade. How do you think companies are leveraging these changes to meet ever changing...
-
Discuss the common sections of grant proposals. [ Tip: Include the Problem Statement / Needs Assessment, Time line or time table, Evaluation, Budget and Budget justification and Letter of Support. ] ...
-
Spain wanted to hold on to the northern border by populating it with immigrants from Mexico, Spain, and making the Indians Spanish citizens, also known as Christians. 1- Why would the Indians agree...
-
Decomposition reaction of A is given as: A 100C D+F Researcher's hypothesized the decomposition reaction as per the following mechanism: A k1 B* +C* k2 B* + A D+E* E* k3 B*+F 2B * k4 G Derive the...
-
Discuss the information available from the following techniques in the analysis of inorganic pigments used in antique oil paintings: (i) Powder X-ray diffraction, (ii) Infrared and Raman...
-
As its name implies, NaHA is an "acid salt" because it has a proton available to donate to a base. Briefly explain why a pH calculation for a solution of NaHA differs from that for a weak acid of the...
-
The nitrobenzene in 300 mg of an organic mixture was reduced to phenylhydroxylamine at a constant potential of 20.96 V (versus SCE) applied to a mercury cathode: C6H5NO2 + 4H+ + 4e ( C6H5NHOH + H2O...
-
The ethyl acetate concentration in an alcoholic solution was determined by diluting a 10.00-mL sample to 100.00 mL. A 20.00-mL portion of the diluted solution was refluxed with 40.00 mL of 0.04672 M...
-
What are the differences among an onsite team, a virtual team, a task force, and a committee? What are some of the potential differences in dynamics between people in these different groups?
-
Compare and contrast disciplinary, interdisciplinary, and crossfunctional teams.
-
What are some of the unique challenges associated with teamwork in health care? How do you see teamwork fitting in with the accountable care organization (ACO) mandates? Describe three benefits and...

Study smarter with the SolutionInn App


