Balance the following acidic skeleton half reaction: Cr07 (aq) Cr+ (aq) A) Enter the coefficient...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
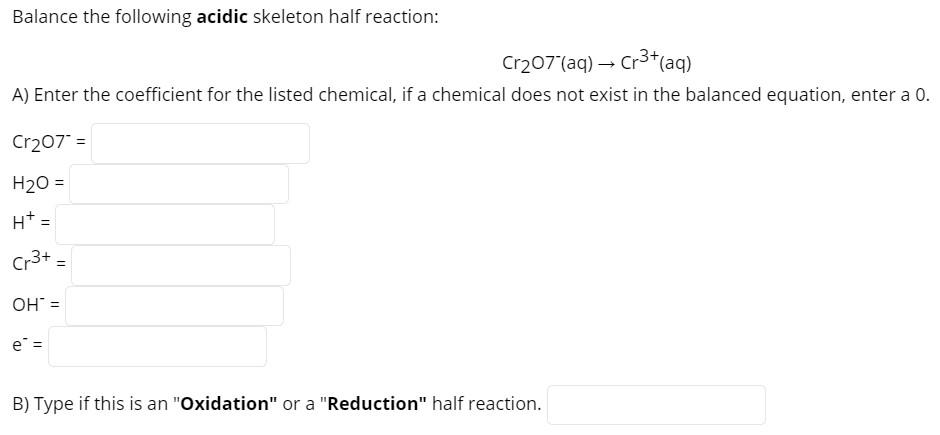
Balance the following acidic skeleton half reaction: Cr₂07 (aq) → Cr³+ (aq) A) Enter the coefficient for the listed chemical, if a chemical does not exist in the balanced equation, enter a 0. Cr207" = H₂O = H* = Cr3+ OH = e = B) Type if this is an "Oxidation" or a "Reduction" half reaction. = Balance the following acidic skeleton half reaction: Cr₂07 (aq) → Cr³+ (aq) A) Enter the coefficient for the listed chemical, if a chemical does not exist in the balanced equation, enter a 0. Cr207" = H₂O = H* = Cr3+ OH = e = B) Type if this is an "Oxidation" or a "Reduction" half reaction. =
Expert Answer:
Answer rating: 100% (QA)
CHOHaq CrO aq CHOaq Cr aq Oxidation Half Reaction CH3OH CH... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
A theoretically correct measure of income does not exist in the real world in which accountants must operate. Required a. What is meant by the phrase a theoretically correct measure of income? b. Why...
-
The hydride ion does not exist in water because it has a greater attraction for the hydronium ion than the hydroxide ion does. Write the equation for the reaction that occurs when calcium hydride is...
-
U.S. requirement of reporting comprehensive income does not exist in the United Kingdom, as the WPP Group does not report comprehensive income anywhere in these statements. However, WPP Group does...
-
Consider the pistoncylinder arrangement shown in the sketch below. The gas forces on each side of the piston, assuming there are no frictional forces at the pistoncylinder interface, balance the...
-
Wisconsin Snowmobile Corp. is considering a switch to level production. Cost efficiencies would occur under level production, and aftertax costs would decline by $36,000, but inventory would increase...
-
A person has had $3,790 withheld for income taxes and a final tax liability of $3,600. Would this be a refund or an additional amount due and for what amount?
-
The Minangkabau, a rice-growing society in West Sumatra, Indonesia, is one of the few matrilineal Islamic societies in South Asia. In Minangkabau villages, kinship relations and families are...
-
A partial amortization schedule for a 10-year note payable issued on January 1, 2016, is shown below: Required a. Using a financial statements model like the one shown here, record the appropriate...
-
As part of her year-end responsibilities, Sharon prepared the company's variable costing income statement as follows. Sales Variable expenses: $418,700 COGS $142,200 Operating expenses 23,700 165,900...
-
Antelopes, native to Africa and Asia, range in size from 30 cm to over 180 cm at the shoulder, with most between 90-120 cm. This is related to the humerus length. The data below gives the length and...
-
f(x+h)-f(x) K For the function f(x)=3x+5, construct and simplify the difference quotient h f(x+h)- f(x) h (Simplify your answer. Use integers or fractions for any numbers in the expression.)
-
To manage this interest rate risk, the FI's manager considers using futures contracts. The manager looks at Treasury note futures, where the duration of the Treasury notes underlying the futures...
-
During the months of November and December 2014, Comfy Home had the following transactions related to holiday candles: November 1, 2014 purchased 200 candles for $5 each Between November 2-30, 2014 ...
-
Given the 80% learning rate, What is the total contract price for the 8 satellites? (Include the 25% markup on cost in the contract price)
-
Goering Eye Care is a small firm with excellent growth opportunities. Goering's sales in 2017 were $2 million. Sales are expected to grow 10% in 2018. At the end of 2017 Goering's total assets were...
-
Q-1 Given that ELC's discount rate is 0.7 percent, what is the present value of future lease payments upon lease commencement? (in thousands of dollars) $760 $777 $1,001 $870
-
Part I: Answer the following questions 1. There are several strategies to persuade your audience. Discuss one strategy in detail and provide an example of how you could use this strategy to persuade...
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
The first step in the preparation of lead from its ore (galena, PbS) consists of roasting the ore. 2PbS(s) + 3O2(g) 2SO2(g) + 2PbO(s) Calculate the standard enthalpy change for this reaction, using...
-
Consider two flasks that contain different pure liquids at 20C. The liquid in one flask, substance A, has a molar mass of 100 g/mol and has hydrogen bonding. The liquid in the other flask, substance...
-
Ozone decomposes to oxygen gas. 2O3(g) 3O2(g) A proposed mechanism for this decomposition is What is the rate law derived from this mechanism? O3 2 + O (fast, equilibrium) slow)
-
How closely related do you believe language and thought to be? During the production of Chapter 3 in this book, the authors and editors debated whether we should use the word "autistics" or the...
-
When did human language originate?
-
Describe American styles of nonverbal communication. Are there different styles of nonverbal communication in different areas of the United States?

Study smarter with the SolutionInn App


