Based on the activity series, which one of the reactions below will occur? A) Pb (s)...
Fantastic news! We've Found the answer you've been seeking!
Question:
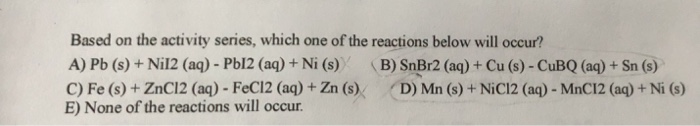
Transcribed Image Text:
Based on the activity series, which one of the reactions below will occur? A) Pb (s) + Nil2 (aq) - Pb12 (aq) + Ni (s) C) Fe (s) + ZnCl2 (aq) - FeC12 (aq) + Zn (s) E) None of the reactions will occur. (B) SnBr2 (aq) + Cu (s) - CuBQ (aq) + Sn (s) D) Mn (s) + NiC12 (aq) - MnC12 (aq) + Ni (s) Based on the activity series, which one of the reactions below will occur? A) Pb (s) + Nil2 (aq) - Pb12 (aq) + Ni (s) C) Fe (s) + ZnCl2 (aq) - FeC12 (aq) + Zn (s) E) None of the reactions will occur. (B) SnBr2 (aq) + Cu (s) - CuBQ (aq) + Sn (s) D) Mn (s) + NiC12 (aq) - MnC12 (aq) + Ni (s)
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Ans B D will occurs Explanation ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Based on energy considerations, which of the following reactions will occur more readily? (a) Cl(g) + CH4(g) CH3Cl(g) + H(g) (b) Cl(g) + CH4(g) CH3(g) + HCl(g)
-
Will damped oscillations occur for any values of b and k? Explain.
-
Below are a series of transactions which are independent of each other. (a) A government makes a $10-million loan to a manufacturer, repayable in 5 years with an interest rate of 2%. Current...
-
Assuming @Weather is a valid repeatable annotation that takes a String, with its associated containing type annotation @Forecast, which of the following can be applied to a type declaration? (Choose...
-
The Cada Corporation is authorized to issue 10,000 shares of $100 par, convertible, callable preferred stock and 80,000 shares of no-par, no-stated-value common stock . There are currently 7,000...
-
Salicylic acid, C6H4OHCOOH, is used in the manufacture of acetylsalicylic acid (aspirin) and methyl salicylate (wintergreen flavor). A saturated solution of salicylic acid contains 2.2 g of the acid...
-
Question: Whitfield, who was black, worked for Ohio Edison. Edison fired him, but then later offered to rehire him. Another employee argued that Edison's original termination of Whitfield had been...
-
Proration of overhead. The Ride-On-Wave Company (ROW) produces a line of non-motorized boats. ROW uses a normal-costing system and allocates manufacturing overhead using direct manufacturing labor...
-
Provincial Income Tax Hemal Thomas is an employee in British Columbia. The net pay calculation is for the first biweekly pay of 2019. The employee is 32 years old and in this pay is entitled to 70...
-
1. What is your assessment of the financial performance of Nelson Nurseries? 2. Do you agree with Christine Barton?s accounts-payable policy? 3. What explains the erosion of the cash balance? 4. What...
-
Your firm is contemplating the purchase of a new $500,000 computer-based order entry system. The system will be depreciated straight-line to zero over its five-year life. It will be worth $44,000 at...
-
The following output from MINITAB presents a confidence interval for the mean difference between matched pairs. a. What is the point estimate of d? b. How many degrees of freedom are there? c. Fill...
-
What is involved in a responsibility reporting system?
-
How does contribution margin differ from controllable margin in a responsibility report for a profit centre?
-
How can the break-even point be determined?
-
What is the difference between direct fixed costs and indirect fixed costs relative to a responsibility report for a profit centre? Which of these costs are called traceable costs and which are...
-
Devise a comprehensive plan to educate parents and guardians about the signs of child maltreatment. What would be important information to include? What resources would be helpful to provide to the...
-
Audrey purchases a riding lawnmower using a 2-year, no-interest deferred payment plan at Lawn Depot for x dollars. There was a down payment of d dollars and a monthly payment of m dollars. Express...
-
The molecules Have the same molecular formula (C3H8O) but different normal boiling points, as shown. Rationalize the difference in boiling points? (b) Ethyl methyl ether (a) Propanol 97.2C 10.8 C
-
Consider the two waves shown here, which we will consider to represent two electromagnetic radiations: (a) What is the wavelength of wave A? Of wave B? (b) What is the frequency of wave A? Of wave B?...
-
The coordination number for the AI3+ ion is typically between four and six. Use the anion coordination number to determine the coordination number in the following compounds: (a) AlF3 where the...
-
Junior Gonzales Racing Fuel is considering the purchase of a fuel truck that he would use to sell gasoline at motor sport racing events in Puerto Rico. He has determined that a used truck is...
-
Karen Calloway is considering adding a new style of gym shorts to her product line. She has determined that it would require an investment of $22,000 to add the new style shorts. She believes that...
-
BRV Construction Company is contemplating the purchase of scaffolding at the cost of \($32,000\). Currently, the company rents similar scaffolding for use at each of its construction sites. The...

Study smarter with the SolutionInn App


