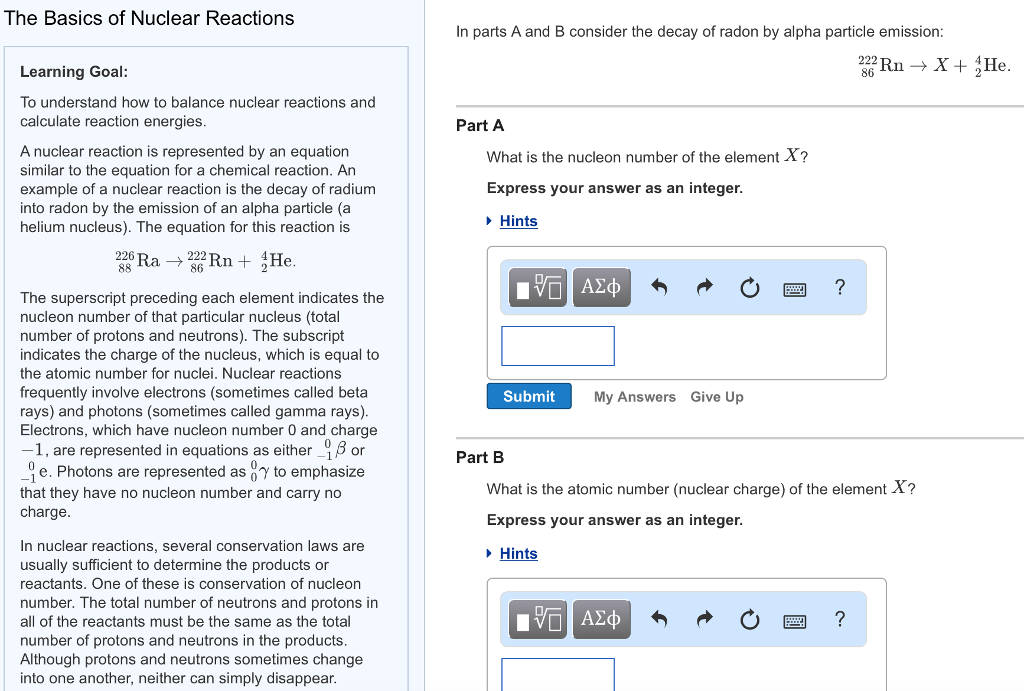
The Basics of Nuclear Reactions Learning Goal: To understand how to balance nuclear reactions and calculate...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
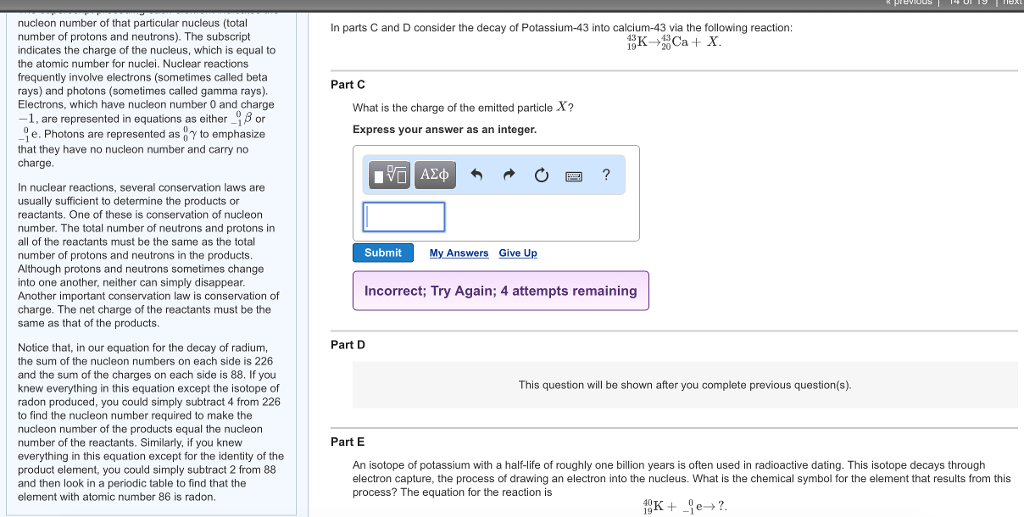
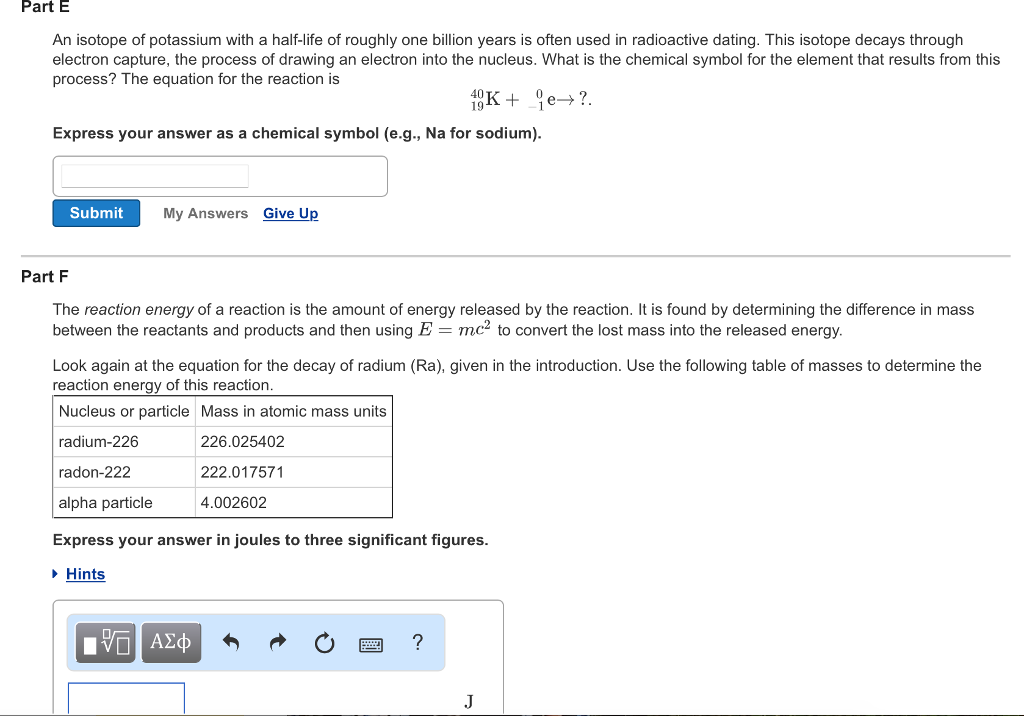
The Basics of Nuclear Reactions Learning Goal: To understand how to balance nuclear reactions and calculate reaction energies. A nuclear reaction is represented by an equation similar to the equation for a chemical reaction. An example of a nuclear reaction is the decay of radium into radon by the emission of an alpha particle (a helium nucleus). The equation for this reaction is 226 Ra → Rn+ He. 88 86 The superscript preceding each element indicates the nucleon number of that particular nucleus (total number of protons and neutrons). The subscript indicates the charge of the nucleus, which is equal to the atomic number for nuclei. Nuclear reactions frequently involve electrons (sometimes called beta rays) and photons (sometimes called gamma rays). Electrons, which have nucleon number 0 and charge -1, are represented in equations as either or 96 e. Photons are represented as to emphasize that they have no nucleon number and carry no charge. In nuclear reactions, several conservation laws are usually sufficient to determine the products or reactants. One of these is conservation of nucleon number. The total number of neutrons and protons in all of the reactants must be the same as the total number of protons and neutrons in the products. Although protons and neutrons sometimes change into one another, neither can simply disappear. In parts A and B consider the decay of radon by alpha particle emission: 222 Rn → X + He. Part A What is the nucleon number of the element X? Express your answer as an integer. ▸ Hints Submit ΑΣΦ ■ My Answers Give Up Part B What is the atomic number (nuclear charge) of the element X? Express your answer as an integer. ▸ Hints X ? ΑΣΦ ? nucleon number of that particular nucleus (total number of protons and neutrons). The subscript indicates the charge of the nucleus, which is equal to the atomic number for nuclei. Nuclear reactions frequently involve electrons (sometimes called beta rays) and photons (sometimes called gamma rays). Electrons, which have nucleon number 0 and charge -1, are represented in equations as either or e. Photons are represented as to emphasize that they have no nucleon number and carry no charge. In nuclear reactions, several conservation laws are usually sufficient to determine the products or reactants. One of these is conservation of nucleon number. The total number of neutrons and protons in all of the reactants must be the same as the total number of protons and neutrons in the products. Although protons and neutrons sometimes change into one another, neither can simply disappear. Another important conservation law is conservation of charge. The net charge of the reactants must be the same as that of the products. Notice that, in our equation for the decay of radium, the sum of the nucleon numbers on each side is 226 and the sum of the charges on each side is 88. If you knew everything in this equation except the isotope of radon produced, you could simply subtract 4 from 226 to find the nucleon number required to make the nucleon number of the products equal the nucleon number of the reactants. Similarly, if you knew everything in this equation except for the identity of the product element, you could simply subtract 2 from 88 and then look in a periodic table to find that the element with atomic number 86 is radon. In parts C and D consider the decay of Potassium-43 into calcium-43 via the following reaction: 43 19K Ca + X. 19¹ Part C What is the charge of the emitted particle X? Express your answer as an integer. 177 ΑΣΦΑ Submit My Answers Give Up Part D P ? Incorrect; Try Again; 4 attempts remaining This question will be shown after you complete previous question(s). previous 14 0 19 Text Part E An isotope of potassium with a half-life of roughly one billion years is often used in radioactive dating. This isotope decays through electron capture, the process of drawing an electron into the nucleus. What is the chemical symbol for the element that results from this process? The equation for the reaction is 19K+ je→?. Part E An isotope of potassium with a half-life of roughly one billion years is often used in radioactive dating. This isotope decays through electron capture, the process of drawing an electron into the nucleus. What is the chemical symbol for the element that results from this process? The equation for the reaction is e?. 10K+ Express your answer as a chemical symbol (e.g., Na for sodium). Submit My Answers Give Up Part F The reaction energy of a reaction is the amount of energy released by the reaction. It is found by determining the difference in mass between the reactants and products and then using E = mc² to convert the lost mass into the released energy. Look again at the equation for the decay of radium (Ra), given in the introduction. Use the following table of masses to determine the reaction energy of this reaction. Nucleus or particle Mass in atomic mass units radium-226 226.025402 radon-222 222.017571 alpha particle 4.002602 Express your answer in joules to three significant figures. ▸ Hints 15. ΑΣΦ ? J The Basics of Nuclear Reactions Learning Goal: To understand how to balance nuclear reactions and calculate reaction energies. A nuclear reaction is represented by an equation similar to the equation for a chemical reaction. An example of a nuclear reaction is the decay of radium into radon by the emission of an alpha particle (a helium nucleus). The equation for this reaction is 226 Ra → Rn+ He. 88 86 The superscript preceding each element indicates the nucleon number of that particular nucleus (total number of protons and neutrons). The subscript indicates the charge of the nucleus, which is equal to the atomic number for nuclei. Nuclear reactions frequently involve electrons (sometimes called beta rays) and photons (sometimes called gamma rays). Electrons, which have nucleon number 0 and charge -1, are represented in equations as either or 96 e. Photons are represented as to emphasize that they have no nucleon number and carry no charge. In nuclear reactions, several conservation laws are usually sufficient to determine the products or reactants. One of these is conservation of nucleon number. The total number of neutrons and protons in all of the reactants must be the same as the total number of protons and neutrons in the products. Although protons and neutrons sometimes change into one another, neither can simply disappear. In parts A and B consider the decay of radon by alpha particle emission: 222 Rn → X + He. Part A What is the nucleon number of the element X? Express your answer as an integer. ▸ Hints Submit ΑΣΦ ■ My Answers Give Up Part B What is the atomic number (nuclear charge) of the element X? Express your answer as an integer. ▸ Hints X ? ΑΣΦ ? nucleon number of that particular nucleus (total number of protons and neutrons). The subscript indicates the charge of the nucleus, which is equal to the atomic number for nuclei. Nuclear reactions frequently involve electrons (sometimes called beta rays) and photons (sometimes called gamma rays). Electrons, which have nucleon number 0 and charge -1, are represented in equations as either or e. Photons are represented as to emphasize that they have no nucleon number and carry no charge. In nuclear reactions, several conservation laws are usually sufficient to determine the products or reactants. One of these is conservation of nucleon number. The total number of neutrons and protons in all of the reactants must be the same as the total number of protons and neutrons in the products. Although protons and neutrons sometimes change into one another, neither can simply disappear. Another important conservation law is conservation of charge. The net charge of the reactants must be the same as that of the products. Notice that, in our equation for the decay of radium, the sum of the nucleon numbers on each side is 226 and the sum of the charges on each side is 88. If you knew everything in this equation except the isotope of radon produced, you could simply subtract 4 from 226 to find the nucleon number required to make the nucleon number of the products equal the nucleon number of the reactants. Similarly, if you knew everything in this equation except for the identity of the product element, you could simply subtract 2 from 88 and then look in a periodic table to find that the element with atomic number 86 is radon. In parts C and D consider the decay of Potassium-43 into calcium-43 via the following reaction: 43 19K Ca + X. 19¹ Part C What is the charge of the emitted particle X? Express your answer as an integer. 177 ΑΣΦΑ Submit My Answers Give Up Part D P ? Incorrect; Try Again; 4 attempts remaining This question will be shown after you complete previous question(s). previous 14 0 19 Text Part E An isotope of potassium with a half-life of roughly one billion years is often used in radioactive dating. This isotope decays through electron capture, the process of drawing an electron into the nucleus. What is the chemical symbol for the element that results from this process? The equation for the reaction is 19K+ je→?. Part E An isotope of potassium with a half-life of roughly one billion years is often used in radioactive dating. This isotope decays through electron capture, the process of drawing an electron into the nucleus. What is the chemical symbol for the element that results from this process? The equation for the reaction is e?. 10K+ Express your answer as a chemical symbol (e.g., Na for sodium). Submit My Answers Give Up Part F The reaction energy of a reaction is the amount of energy released by the reaction. It is found by determining the difference in mass between the reactants and products and then using E = mc² to convert the lost mass into the released energy. Look again at the equation for the decay of radium (Ra), given in the introduction. Use the following table of masses to determine the reaction energy of this reaction. Nucleus or particle Mass in atomic mass units radium-226 226.025402 radon-222 222.017571 alpha particle 4.002602 Express your answer in joules to three significant figures. ▸ Hints 15. ΑΣΦ ? J
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
A LTI system is represented by an ordinary differential equation (a) Obtain the transfer function H(s) = Y(s)/X(s) = B(s)/A(s) and find its poles and zeros. Is this system BIBO stable? Is there any...
-
A first-order system is represented by the time domain differential equation A feedback controller is to be designed such that u(t) = -kx, and the desired equilibrium condition is x(t) = 0 as t ->...
-
A nonlinear device is represented by the function y = f(x) = ex, where the operating point for the input x is x0 = 1. Determine a linear approximation valid near the operating point.
-
What is the result of executing the following method? A. The declaration of name does not compile. B. The declaration of _number does not compile. C. The declaration of profit$$$ does not compile. D....
-
John Wilson, a portfolio manager, is evaluating the expected performance of two common stocks, Furhman Labs, Inc., and Garten Testing, Inc. The risk-free rate is 5 percent, the expected return on the...
-
Describe the operation of the prompt method.
-
To calculate the expected frequencies, we must know the row totals, the column totals, and the ________________ total. In Exercises 35, fill in each blank with the appropriate word or phrase.
-
Suppose the current USD/euro exchange rate is 1.2000 dollar per euro. The six month forward exchange rate is 1.1950. The six month USD interest rate is 1% per annum continuously compounded? Estimate...
-
In a marginal costing system only variable costs would be assigned to products or services, in which case management may rely on a contribution approach to decisions. REQUIRED : Explain and discuss...
-
Lets return to the model of financial aid awards at a liberal arts college that was first introduced in Section 2.2. In that section, we estimated the following equation (standard errors in...
-
Interpret the following situations : a. Jennifer just received the results of her SAT exam. Her SAT Mathematics score of 600 is in the 74th percentile. What does this mean? b. Time taken to finish a...
-
If 6 of 18 new buildings in a city violate the building code, what is the probability that a building inspector, who randomly selects 4 of the new buildings for inspection, will catch (a) none of the...
-
Why do sample surveys have predetermined margins of error?
-
Nan-Clean is a newly developed cleaning liquid for milk bottles that uses special enzymes to remove dirt effectively. Nan-Clean has been positioned as a product that is made of natural ingredients...
-
How does laddering as a technique help in qualitative research?
-
A newly established noodle house has suffered low sales lately. The owner of the noodle house wants to research to collect information that will help him solve the problem at hand and design a...
-
A company just issued a bond that will pay the bearer $200,000 in fifteen years. As this is a bearer instrument, there are no interim interest or coupon payments. a. If 5,000 of these bonds were sold...
-
In your audit of Garza Company, you find that a physical inventory on December 31, 2012, showed merchandise with a cost of $441,000 was on hand at that date. You also discover the following items...
-
The following problems relate to periodicity and power of discrete-time signals. (a) Is the signal x[n] = e j(n8)/8 periodic? if so determine its fundamental period N 0 . What if x 1 [n] = e...
-
The output of a discrete-time system is y[n] = w[n] x[n] where x[n] is the input, and w[n] = u[n] u[n 5] is a rectangular window. (a) The input is x[n] = 4 sin (n/2), < n < , determine if x[n] is...
-
If we wish to preserve low frequencies components of the input, a low-pass Butterworth filter could perform better than a Chebyshev filter. MATLAB provides a second Chebyshev filter function cheby2...
-
Assume Knowlton Holdings Ltd. completed these long-term non-strategic investment transactions during 2020 : 1. Journalize Knowlton's investment transactions assuming the company reports any changes...
-
Suppose on February 1, 2020, General Motors paid \(\$ 41\) million for a \(40 \%\) investment in ABC Ltd., an auto parts manufacturer. Assume \(A B C\) earned net income of \(\$ 6\) million and paid...
-
Return to exercise SB-7, the Prudential Bache (PB) investment in CitiCorp bonds. Journalize the following on PB's books: a. Purchase of the bond investment on January 2, 2020. PB expects to hold the...

Study smarter with the SolutionInn App


