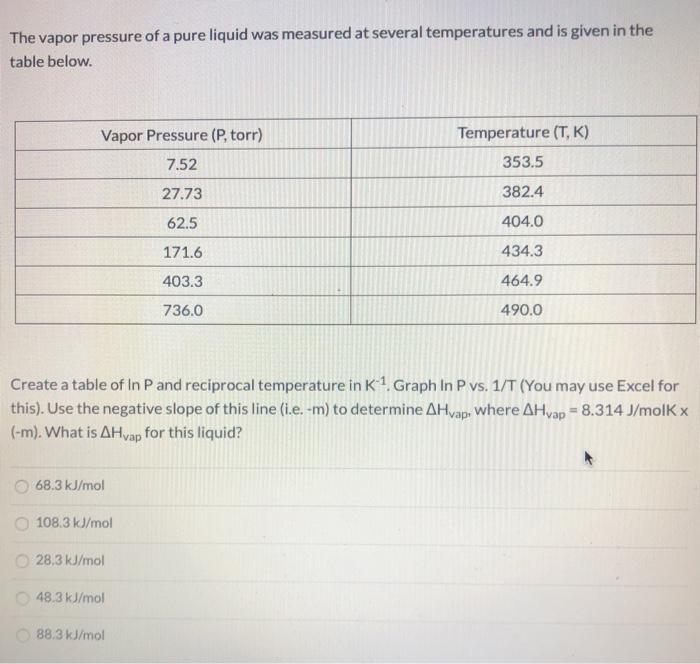
The vapor pressure of a pure liquid was measured at several temperatures and is given in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The vapor pressure of a pure liquid was measured at several temperatures and is given in the table below. Vapor Pressure (P, torr) Temperature (T, K) 7.52 353.5 27.73 382.4 62.5 404.0 171.6 434.3 403.3 464.9 736.0 490.0 Create a table of In P and reciprocal temperature in K1. Graph In P vs. 1/T (You may use Excel for this). Use the negative slope of this line (i.e. -m) to determine AHvap, Where AHvap = 8.314 J/molK x (-m). What is AHvap for this liquid? 68.3 kJ/mol 108.3 kJ/mol 28,3 kJ/mol O 48.3 kJ/mol O 88.3 kJ/mol The vapor pressure of a pure liquid was measured at several temperatures and is given in the table below. Vapor Pressure (P, torr) Temperature (T, K) 7.52 353.5 27.73 382.4 62.5 404.0 171.6 434.3 403.3 464.9 736.0 490.0 Create a table of In P and reciprocal temperature in K1. Graph In P vs. 1/T (You may use Excel for this). Use the negative slope of this line (i.e. -m) to determine AHvap, Where AHvap = 8.314 J/molK x (-m). What is AHvap for this liquid? 68.3 kJ/mol 108.3 kJ/mol 28,3 kJ/mol O 48.3 kJ/mol O 88.3 kJ/mol
Expert Answer:
Answer rating: 100% (QA)
Select a middle data and Apply Clausius Clapeyron equation ... View the full answer

Related Book For 

Practicing Statistics Guided Investigations for the Second Course
ISBN: 978-0321586018
1st edition
Authors: Shonda Kuiper, Jeff Sklar
Posted Date:
Students also viewed these accounting questions
-
In Section 11.5 we defined the vapor pressure of a liquid in terms of an equilibrium. (a) Write the equation representing the equilibrium between liquid water and water vapor and the corresponding...
-
The data given in the table below are the midterm scores in a course for a sample of 10 students and the scores of student evaluations of the instructor. (In the instructor evaluation scores, 1 is...
-
The following table gives the vapor pressure of hexafluorobenzene (C6F6) as a function of temperature: (a) By plotting these data in a suitable fashion, determine whether the Clausius-Clapeyron...
-
X-Tech Inc. produces specialized bolts for the aerospace industry. The operating cost of producing a single bolt is $2. The company currently sells the bolts for $6/unit. Each time the company...
-
For the data in Exercise, In Exercise At a major credit card bank, the percentages of people who historically apply for the Silver, Gold, and Platinum cards are 60%, 30%, and 10%, respectively. In a...
-
A solar collector is placed in direct sunlight where it absorbs energy at the rate of 880 J/s for each square meter of its surface. The emissivity of the solar collector is e = 0.75. What equilibrium...
-
Consider the National Football League data in Table B.1. Build a linear regression model relating the number of games won to the yards gained rushing by opponents $x_{8}$, the percentage of rushing...
-
Ajax Corporation has two divisions. The Mining Division makes toldine, which is then transferred to the Metals Division. The toldine is further processed by the Metals Division and sold to customers...
-
The following information was reported in the December 31 financial statements of National Airways, Incorporated (listed alphabetically, amounts in millions). Accounts Payable Accounts Receivable...
-
Jim and Joel have come to you to get an appraisal to lease their property. Using the information below, complete the following questions: Answer the below questions according to the property details...
-
A firm is considering investing in a particular project and has asked its capital budgeting section to evaluate that project. After collecting information from various sources, the following...
-
Suppose the expected return on the market is 14%, market volatility is 27%, and the risk-free rate is 2%. a. Suppose stock A has a beta of 1.25. According to the CAPM, what is its expected return?...
-
A startup has 1,000 shares outstanding. A VC offers to invest $300,000 at a pre-money price of $1,500 per share. What is the pre-money valuation? Give your answer in millions of dollars (for example,...
-
A startup has 1,000 shares outstanding. A VC offers to invest $500,000 at a pre-money valuation of $800,000. What is the post-money valuation? Give your answer in millions of dollars (for example, if...
-
Tuffy Corp. has the following normal account balances on 12/31/X1 before the year-end determination of its inventory cost using the dollar-value LIFO (DVL) method: Account Balance on 12/31/X1:...
-
You work for a company that is evaluating developing a new online video game. Your company will need to spend $2 million per year in software development costs at the end of each of the next four...
-
Provide a supply and demand graph to show impact of following on equilibrium prices and quantity for texas instrument calculator as well as filling in the blanks. Thanks! Question 12 Use a supply and...
-
What does non-recourse financing mean?
-
Use software to calculate the eigenvectors of R.
-
In 2009, three Grinnell students (Allie Greenberg, Hannah Lytle, and Phillip Brogdon) conducted an analysis to estimate the probability of winning the Academy Award for Best Picture. The Academy...
-
State the null and alternative hypotheses for this study. Conduct an ANOVA to analyze the Flower data. Check the normality and equal variance assumptions. State your conclusions based on the p-...
-
What is the relation between degrees Fahrenheit and degrees Rankine? And the relation between degrees Celsius and Kelvin?
-
State Newton's second law as you would apply it to a control mass.
-
Define a 1-pound force in terms of the acceleration it will give to a 1-pound mass. Give a similar definition for a newton in the SI system.

Study smarter with the SolutionInn App


