7. Calculate the volumetric flow of water at 5 ? C through the system illustrated in the...
Fantastic news! We've Found the answer you've been seeking!
Question:
7. Calculate the volumetric flow of water at 5 ? C through the system illustrated in the figure.
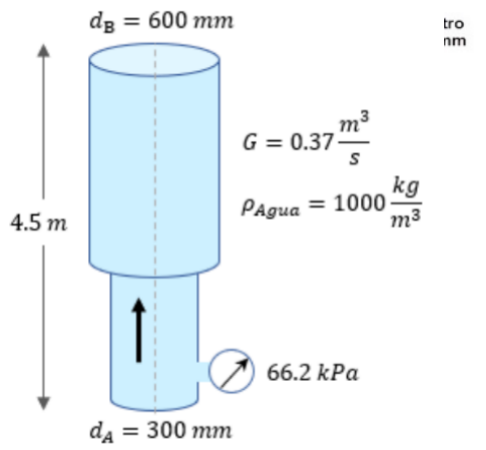
Transcribed Image Text:
dg : 3D 600 тm tro nm G = 0.37- kg PAgua = 1000- m3 4.5 m ↑ 66.2 kPa da = 300 mm %3D dg : 3D 600 тm tro nm G = 0.37- kg PAgua = 1000- m3 4.5 m ↑ 66.2 kPa da = 300 mm %3D
Expert Answer:
Answer rating: 100% (QA)
ha Om h 45m 1m da 300mm 03m 1000mm 1m dB 600mm 06m 1000mm PA ... View the full answer

Posted Date:
Students also viewed these physics questions
-
Derive the expression to calculate the volumetric flow rate from a porous media using Darcy Equation for radial and parallel flow system. Hence drive the expression to calculate the average...
-
Given is steady isothermal flow of water at 20°C through the device in Fig. P3.126. Heat-transfer, gravity, and temperature effects are negligible. Known data are D1 = 9 cm, Q1 = 220 m3/h, p1 =...
-
What is the specific volume of water at 5 MPa and 100C? What would it be if the incompressible liquid approximation were used? Determine the accuracy of this approximation.
-
Jefferson Company purchased a piece of equipment on January 1, 2014. The equipment cost $60,000 and has an estimated life of 8 years and a salvage value of $8,000. What was the depreciation expense...
-
Repeat Prob. 4.4-3 for the following problem. Maximize Z = 2x1 + 3x2, Subject to and x1 ¥ 0, x2 ¥ 0. xi 2x2 s 30 1
-
For an 85 wt% Pb-15 wt% Mg alloy, make schematic sketches of the microstructure that would be observed for conditions of very slow cooling at the following temperatures: 600C (1110F), 500C (930F),...
-
In his 2023 State of the Union address, President Joe Biden stated, As I stand here tonight, we have created a record 12 million new jobs. Is it likely that the U.S. economy created only 12 million...
-
Control Enterprises produces touch screens for use in various smartphones. The company reports the following information at December 31. Control Enterprises began operations on January 31 earlier...
-
Question 11pts The APRN unit director is working at an agency that has a high incidence of medication errors, specifically with heparin. One recent error had a patient receive twice the prescribed...
-
A project consists of 10 activities, lettered A through J, as shown in the accompanying precedence diagram, with the activities on the arrows. For each activity, the deterministic time estimate in...
-
Please do fast Find the integrals using complex analysis + 1 (2+cose)
-
Consider the following statements regarding a linear system \(y=f(x)\) : 1. \(f\left(x_{1}+x_{2} ight)=f\left(x_{1} ight)+f\left(x_{2} ight)\) 2. \(f[x(t+T)]=f[x(t)]+f[x(T)]\) 3. \(f(K x)=K f(x)\) Of...
-
How many three-digit numbers can be formed using the numbers \(\{1,2,3,4,5,6,7\}\) without repetition?
-
How are operations related to messages?
-
Traveling on, you meet Merlin and Meredith. Merlin says, "If I am a truth teller, then Meredith is a truth teller." Is Merlin a liar or a truth teller? What about Meredith? Explain your answer. In...
-
A linear system, initially at rest, is subjected to an input signal;.............. \[ r(t)=1-e^{-t}(t \geq 0) \] The response of the system for \(t \geq 0\) is given by \[ c(t)=1-e^{-2 t} \] The...
-
GMPK LLP, a firm that provides financial services, has suffered financially due to a marked loss of reputation to the point where it cannot now meet its debts. There are several options applicable to...
-
Q1) What is the a3 Value Q2) What is the a7 Value Q3) What is the a4 Value Q4) What is the b3 Value Q5) What is the b2 Value Q6) What is the sign of 2nd constraint? A pastry chef at a bakery wants to...
-
A number of Latin American countries export coffee and import other goods. A longterm drought now reduces coffee production in the countries of this region. Assume that they remain exporters of...
-
Pugelovias growth has been oriented toward expansion of its export industries. How do you think Pugelovias terms of trade have been changing during this time period?
-
Why does the HeckscherOhlin theory predict that most research and development (R&D) activity is done in the industrialized countries?

Study smarter with the SolutionInn App

