The following information is available for Sheridan Products: Accounts receivable Cost of goods sold Inventory (ending)...
Fantastic news! We've Found the answer you've been seeking!
Question:
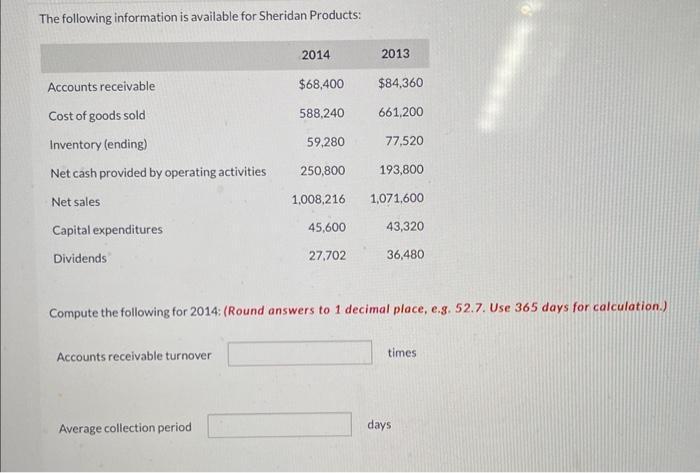
Transcribed Image Text:
The following information is available for Sheridan Products: Accounts receivable Cost of goods sold Inventory (ending) Net cash provided by operating activities Net sales Capital expenditures Dividends Accounts receivable turnover 2014 Average collection period $68,400 588,240 59,280 250,800 1,008,216 45,600 27,702 2013 $84,360 661,200 77,520 193,800 1,071,600 43,320 Compute the following for 2014: (Round answers to 1 decimal place, e.g. 52.7. Use 365 days for calculation.) 36,480 times days The following information is available for Sheridan Products: Accounts receivable Cost of goods sold Inventory (ending) Net cash provided by operating activities Net sales Capital expenditures Dividends Accounts receivable turnover 2014 Average collection period $68,400 588,240 59,280 250,800 1,008,216 45,600 27,702 2013 $84,360 661,200 77,520 193,800 1,071,600 43,320 Compute the following for 2014: (Round answers to 1 decimal place, e.g. 52.7. Use 365 days for calculation.) 36,480 times days
Expert Answer:
Answer rating: 100% (QA)
Accounts receivable turnovernet salesaccounts receivable Accounts rec... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
One of the amino acids in Table 27.1 is the biological precursor to y-aminobutyric acid (4-aminobutanoic acid), which it forms by a decarboxylation reaction. Which amino acid is this?
-
In a statistics class taught by one of the authors, students took their pulses before and after being frightened. The frightening event was having the teacher scream and run from one side of the room...
-
In a management seminar taught by one of the authors to supervisors in a large shipyard, one supervisor commented: We have very little opportunity to motivate employees. All monetary factorsstarting...
-
Density is not routinely measured in the atmosphere. Why is this not a problem for atmospheric scientists?
-
What are the characteristics of a data set that would lead you to construct a pie chart?
-
Calculate Saras profit-maximizing output, price, and economic profit. Web site at which people can design and buy sweatshirts. Sara pays $1,000 a week for her Web server and Internet connection. The...
-
Find a case in California that discusses discovery abuse. What was the abuse? Were there sanctions given by the court against one party for the abuse?
-
The SEC Form 10-K of Google is reproduced online at www.wiley.com/college/pratt. REQUIRED: Review the 10-K, and answer the following questions: a. What are the major sources of cash for the company...
-
1. Yellow Corp. is evaluating an extra dividend versus a share repurchase. In either case, $4,500 would be spent. Current earnings are $0.96 per share and the stock currently sells for $43 per share....
-
? Dean and Ellen Price are married and have a manufacturing business. They bought a piece of business equipment (7-year personal property) on 4/1/2017 for $50,000. Use half-year convention to...
-
Is Methyl Diantilis more odoriferous, more pure, and in better yield than the corresponding vanillyl ethers?
-
Clair is a soccer player that just signed a 3-year contract with a new team. The contract will pay her as follows: $58,000 today $67,000 at the end of year 1 $73,000 at the end of year 2 $89,000 at...
-
Badem Farm issued a 30 year, 6% semi-annual bond 5 years ago. The bond currently sells for $1,040. The company tax rate is 22%. What is the after tax cost of debt?
-
The table shows median annual earnings for women and men with various levels of education. Assuming the difference in the table remains constant over a 40-year career, approximately how much more...
-
7. A NACA 0008 airfoil rapidly changes angle of attack from 3 degrees to 8 degrees. The chord of the airfoil is 0.3 m, the free stream velocity is 10 m/s, and the airfoil is operating in air at...
-
Rousan contributed $1900.00 at the beginning of every month to an RRSP. Interest on the account is 4.95% compounded monthly. If she converted the balance after 13 years into an RRIF paying 7.1%...
-
Questions Project Structure 2 3 4 LO 5 10 6 Circle.java Cylinder.java X public class Circle { 1 related problem Test.java X private double radius; public Circle (double radius) { this.radius =...
-
Big Jim Company sponsored a picnic for employees and purchased a propane grill equipped with a standard-sized propane tank for the picnic. To make sure there was enough propane for all the cooking...
-
Apply the knowledge gained by studying Table 20.3 to help you predict the major organic product of each of the following reactions: (a) Acetic anhydride + ammonia (2 mol) (b) Phthalic anhydride +...
-
Which molecular orbital of ethylene ( or *) is the most important one to look at in a reaction in which ethylene is attacked by an electrophile?
-
Suggest reasonable mechanism for each of the following reactions: (a) (b) (CH3 0 NaOCH 2 CH3OH Cl (88%) (CH3) CCHCHCH,OH NCH CHCH)a Cl
-
The consistent mass matrix of a bar element is given by a. \(\frac{ho A l}{6}\left[\begin{array}{ll}2 & 1 \\ 1 & 2\end{array} ight]\) b. \(\frac{ho A l}{6}\left[\begin{array}{rr}2 & -1 \\ -1 &...
-
How are fixed boundary conditions incorporated in the finite element equations?
-
True or False. The system mass matrix is always singular unless the boundary conditions are incorporated.

Study smarter with the SolutionInn App


