Potable water (total dissolved solids ...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
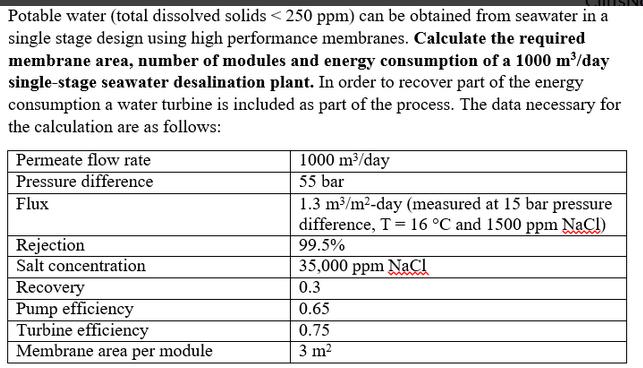
Potable water (total dissolved solids <250 ppm) can be obtained from seawater in a single stage design using high performance membranes. Calculate the required membrane area, number of modules and energy consumption of a 1000 m³/day single-stage seawater desalination plant. In order to recover part of the energy consumption a water turbine is included as part of the process. The data necessary for the calculation are as follows: Permeate flow rate Pressure difference Flux Rejection Salt concentration Recovery Pump efficiency 1000 m³/day 55 bar 1.3 m³/m²-day (measured at 15 bar pressure difference, T 16 °C and 1500 ppm NaCl) 99.5% 35,000 ppm NaCl 0.3 0.65 0.75 Membrane area per module 3 m² Turbine efficiency Potable water (total dissolved solids <250 ppm) can be obtained from seawater in a single stage design using high performance membranes. Calculate the required membrane area, number of modules and energy consumption of a 1000 m³/day single-stage seawater desalination plant. In order to recover part of the energy consumption a water turbine is included as part of the process. The data necessary for the calculation are as follows: Permeate flow rate Pressure difference Flux Rejection Salt concentration Recovery Pump efficiency 1000 m³/day 55 bar 1.3 m³/m²-day (measured at 15 bar pressure difference, T 16 °C and 1500 ppm NaCl) 99.5% 35,000 ppm NaCl 0.3 0.65 0.75 Membrane area per module 3 m² Turbine efficiency
Expert Answer:
Answer rating: 100% (QA)
Calculation of Membrane Area Modules and Energy Consumption 1 Permeate Flow Rate Conversion The permeate flow rate is given in cubic meters per day md... View the full answer

Related Book For 

Understanding Business Statistics
ISBN: 978-1118145258
1st edition
Authors: Stacey Jones, Tim Bergquist, Ned Freed
Posted Date:
Students also viewed these accounting questions
-
Norway has a long tradition of boatbuilding stretching back to before Viking times. This is due, no doubt, to Norways proximity to the sea, ample forests, and long coastline. Among luxury sailing...
-
RTM Technology is an American technology company with several divisions. One division, Sacramento Division, manufactures and sells network components. Sacramento currently sells a single product, an...
-
Key West Electronics, located in Miami, was just ten years old in early 2010, with sales expected to pass the $500 million and net income just beginning to turn positive after years of losses or near...
-
A ball is located 8 0 m above the sea level. Its initial velocity is 1 0 0 m/s and the angle between the horizontal is 37 degrees. How long will it take for the ball to hit the surface? g=9.8m/s 2
-
We are in an age of growing media attention towards trans-identity. What is this? What, specifically, can we learn from Dolezal and Jenner?
-
What additional disclosures are required for trading, available- for-sale, and held-to-maturity securities?
-
When the auditor is unable to confirm accounts receivable, indicate (a) the alternative procedures he may perform and (b) the effects on his audit report.
-
The chemical reactor shown below has a cover (called a head) that is held in place by a series of bolts. The head is made of stainless steel (SG = 8.0), is 3 in. thick, has a diameter of 24 in., and...
-
Don may not be the brightest candle on the cake. He has more taxes withheld from his paycheck so he will get a large check come April from the Federal government. Well, this check came in the mail...
-
Janice Earl is employed at a monthly salary of $700,000. His employer is in the private sector and pays 6HEART trainees. Additionally, Janice contributes $16,000 to Sagicor for his life insurance...
-
ACC-112 M Question 1 Ch 910 1-4-Com X On January 2, 20x1, Valentina x Romheducation.com/ext/map/index.html? con-con&external browser-08launchUrl=https%253A%252F%252Flms.mheducation.com6252Fm Student...
-
Your examination of Procter & Gamble's fiscal 2023 liquidity and current ratio is quite perceptive. It's true that the current ratio being less than 1 indicates P&G's current assets are less than its...
-
Carson wondered if his uncle should consider a different allocation base, since this construction business is both labor-intensive and material-intensive. Charles agrees and determines that total DM...
-
Carol has a bail bondsman post a bail of $10,000 for her trial that will take place in 4 months. The bondsman charges a $80 fee plus of the bail. What interest rate is being charged by the bail...
-
Continue with part (B) above. Assume US tax law allowed USA Logistics to elect not to deduct the $350 million of royalties paid to EuroConnect (i.e., USA Logistics could elect to ignore all of this...
-
Over the years, you developed a friendship with Atieno, the owner-operator of a fast food restaurant. Because of recent government regulations in relation to managing the current pandemic, Atieno has...
-
What is the difference between the straight-line method of depreciation and the written down value method? Which method is more appropriate for reporting earnings?
-
Use the binomial table to find the following probabilities: a. P(x = 6), where n = 11, p = .65 b. P(x > 5), where n = 7, p = .8 c. P(x < 17), where n = 20, p = .7 d. P(9 < x < 12), where n = 16, p =...
-
Samir is selecting stocks for his portfolio. He has 13 candidate stocks from which he intends to select eight. How many different portfolios of eight stocks are possible?
-
A questionnaire concerning the effect of a recent advertising campaign was sent out to a sample of 500 consumers. Results of the study are reported in the cross-tabulation table below: Convert the...
-
Prove that \(C_{P}>C_{V}\), where the notations have their usual meanings.
-
In a chemical process plant, water at 67C is pumped from a storage tank at the rate of 20,000 kg/hr. The motor for the pump expenses work at the rate of 1.5 hp. The water passes through a heat...
-
\(5 \mathrm{~kg}\) of \(\mathrm{N}_{2}\) is heated from an initial state of \(37^{\circ} \mathrm{C}\) and \(101.33 \mathrm{kPa}\) until its temperature reaches \(237^{\circ} \mathrm{C}\). Calculate...

Study smarter with the SolutionInn App


