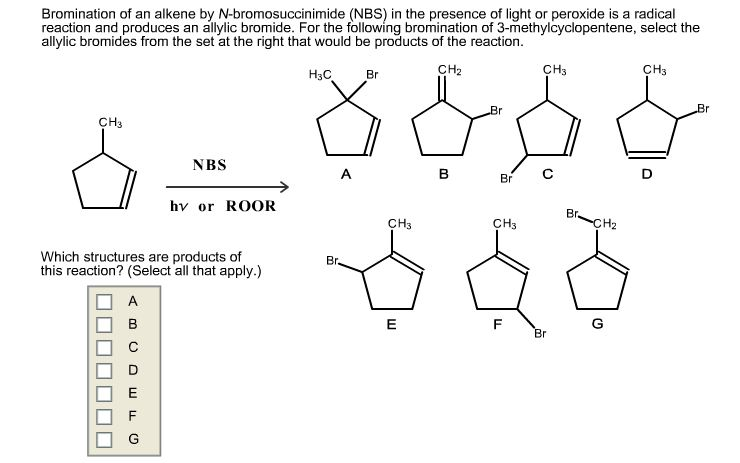
Bromination of an alkene by N-bromosuccinimide (NBS) in thepresence of light or peroxide is a radical reaction
Fantastic news! We've Found the answer you've been seeking!
Question:
Bromination of an alkene by N-bromosuccinimide (NBS) in thepresence of light or peroxide is a radical reaction and produces anallylic bromide. For the following bromination of3-methylcyclopentene, select the allylic bromides from the set atthe right that would be products of the reaction.
Related Book For 

Posted Date:




