BSO, Incorporated, has assets of $740,000 and liabilities of $555,000, resulting in a debt-to-assets ratio of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
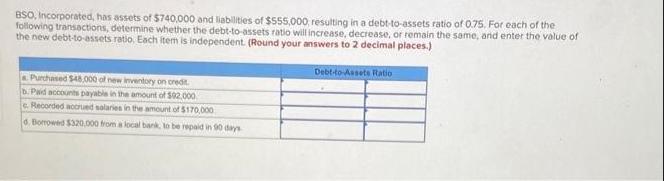
BSO, Incorporated, has assets of $740,000 and liabilities of $555,000, resulting in a debt-to-assets ratio of 0.75. For each of the following transactions, determine whether the debt-to-assets ratio will increase, decrease, or remain the same, and enter the value of the new debt-to-assets ratio. Each item is independent. (Round your answers to 2 decimal places.) & Purchased $48,000 of new inventory on credit. b. Paid accounts payable in the amount of $92,000 c. Recorded accrued salaries in the amount of $170,000 d. Borrowed $320,000 from a local bank, to be repaid in 90 days Debt-to-Assets Ratio BSO, Incorporated, has assets of $740,000 and liabilities of $555,000, resulting in a debt-to-assets ratio of 0.75. For each of the following transactions, determine whether the debt-to-assets ratio will increase, decrease, or remain the same, and enter the value of the new debt-to-assets ratio. Each item is independent. (Round your answers to 2 decimal places.) & Purchased $48,000 of new inventory on credit. b. Paid accounts payable in the amount of $92,000 c. Recorded accrued salaries in the amount of $170,000 d. Borrowed $320,000 from a local bank, to be repaid in 90 days Debt-to-Assets Ratio
Expert Answer:
Answer rating: 100% (QA)
To determine the impact on the debttoassets ratio for each transaction we need to calculate the new ... View the full answer

Related Book For 

Fundamentals of Financial Accounting
ISBN: 978-0078025914
5th edition
Authors: Fred Phillips, Robert Libby, Patricia Libby
Posted Date:
Students also viewed these accounting questions
-
Adria Lopez created Success Systems on October 1, 2013. The company has been successful, and its list of customers has grown. To accommodate the growth, the accounting system is modified to set up...
-
Review the February 26 and March 25 transactions for Business Solutions (SP 4) from Chapter 4. Santana Rey created Business Solutions on October 1, 2016. The company has been successful, and its list...
-
The A. J. Swim Team soon will have an important swim meet with the G. N. Swim Team. Each team has a star swimmer (John and Mark, respectively) who can swim very well in the 100- yard butterfly,...
-
A two block system with weights W and Wo is shown above. Wo is pulling W over the table at a steady velocity U. Derive the algebraic expression for this velocity as it slides on a film of oil with a...
-
We stated in Section 16.4 that MMs proof of dividend irrelevance assumes that new shares are sold at a fair price. Look back at question 4. Assume that new shares are issued in year 1 at $10 a share....
-
Collaboration in a CPFR sense requires much effort from all parties concerned. After your exploration of West Marine, what are some of the hidden problems that come to light after a firm tries to...
-
Name four application controls.
-
Arndt, Inc., reported the following for 2013 and 2014 ($ in millions): a. Expenses each year include $30 million from a two-year casualty insurance policy purchased in 2013 for $60 million. The cost...
-
The following data was compiled from the financial records for the month of May Standard Actual, Direct Materials Price $7.25 $7.46 Direct Materials Quantity 8,000 7,895 Direct Labor Rate $38.00...
-
Consider an ensemble learning algorithm that uses simple majority voting among M learned hypotheses (you may assume M is odd). Suppose that each hypothesis has error & where 0.5 > >0 and that the...
-
Given the geometric sequences below: 6, 36, 216,What is the common ration: r = ______? Find the next two missing terms in the following geometric sequence: _____________, __________
-
3) Eight Senate committees-Appropriations (A), Budget (B), Commerce (C), Energy (D), Environment (E), Foreign Relations (F), Homeland Security and Governmental Affairs (G), and Health (H) need to...
-
Small spiders are known to fly from place to place by letting out a length of silk, which then carries them around. Recently it was discovered that they're actually using the electric field of the...
-
Build an Excel Model to model the problem and answer the following questions (provide amodel template of each question). (a) Use goal programming proposed in the article to formulate this model (b)...
-
INSTRUCTIONS: We're looking to work with a new publisher that is interested in selling us two different placements. The publisher has told us that placement 1 has an approximate CTR of 0.3% and will...
-
Conclusive Remarks 4. Draw overall conclusions and make recommendations based on the evaluation of your chosen organization. 4.1. Summarize the process of evaluating an organization's strategy. 4.2....
-
What's the stock's market value? Do = 2.00, r = 13%, g = 6%. Constant growth model: D Po= rs-8 What is the stock's market value one year from now, P? D will have been paid, so expected dividends are...
-
Frontland Advertising creates, plans, and handles advertising campaigns in a three-state area. Recently, Frontland had to replace an inexperienced office worker in charge of bookkeeping because of...
-
Johnson & Johnson, the maker of Tylenol, uses GAAP. Bayer, the maker of aspirin, uses IFRS. Explain what complications might arise when comparing the Property, Plant, and Equipment of these two...
-
You work for a public company that has relied heavily on debt financing in the past and is now considering a preferred stock issuance to reduce its debt-to-assets ratio. Debt-to-assets is one of the...
-
Describe the specific types of inventory reported by merchandisers and manufacturers.
-
The heat of vaporization of ether is \(25.98 \mathrm{~kJ} / \mathrm{mol}\) at its boiling point, \(34.5^{\circ} \mathrm{C}\). (a) Calculate the rate of change of vapour pressure with temperature...
-
For a gas obeying the equation of state \(V=B+\frac{R T}{P}\), the Joule-Thomson coefficient is given by \[ \mu_{\mathrm{JT}}=-\frac{1}{C_{P}}\left(T \frac{d B}{d T}-B ight) \]
-
Calculate the residual enthalpy and entropy for carbon dioxide at \(393 \mathrm{~K}\) and \(12 \mathrm{MPa}\) using any equation of state.

Study smarter with the SolutionInn App


