Question: Buffer Preparation: Acid-Base Reactions An aqueous solution contains 0.496 M hydrocyanic acid. Calculate the pH of the solution after the addition of 2.18x10 2
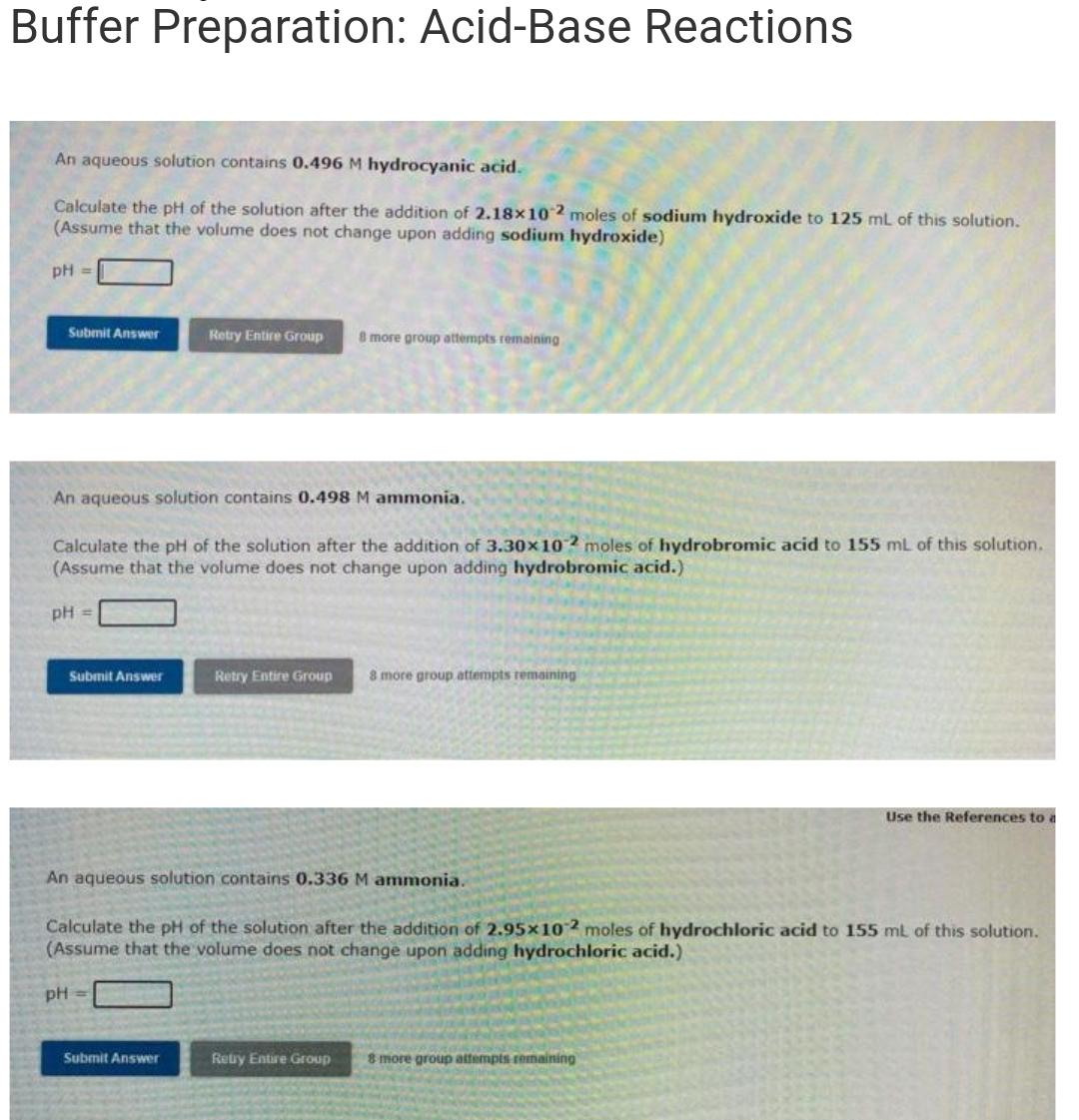
Buffer Preparation: Acid-Base Reactions An aqueous solution contains 0.496 M hydrocyanic acid. Calculate the pH of the solution after the addition of 2.18x10 2 moles of sodium hydroxide to 125 mL of this solution. (Assume that the volume does not change upon adding sodium hydroxide) pH = Submit Answer Retry Entire Group 8 more group attempts remaining An aqueous solution contains 0.498 M ammonia. Calculate the pH of the solution after the addition of 3.30x102 moles of hydrobromic acid to 155 mL of this solution. (Assume that the volume does not change upon adding hydrobromic acid.) pH = Submit Answer Retry Entire Group 8 more group attempts remaining Use the References to a An aqueous solution contains 0.336 M ammonia. Calculate the pH of the solution after the addition of 2.95x102 moles of hydrochloric acid to 155 mL of this solution. (Assume that the volume does not change upon adding hydrochloric acid.) pH = Submit Answer Retry Entire Group 8 more group attempts remaining
Step by Step Solution
3.38 Rating (145 Votes )
There are 3 Steps involved in it
THCNJ 0496 M moles of NaoH added of solution Volume NaoH No of moles of Na... View full answer

Get step-by-step solutions from verified subject matter experts


