By overlying THREE two-dimensional layers of specifically packed atoms (around 20 atoms per layer), please demonstrate...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
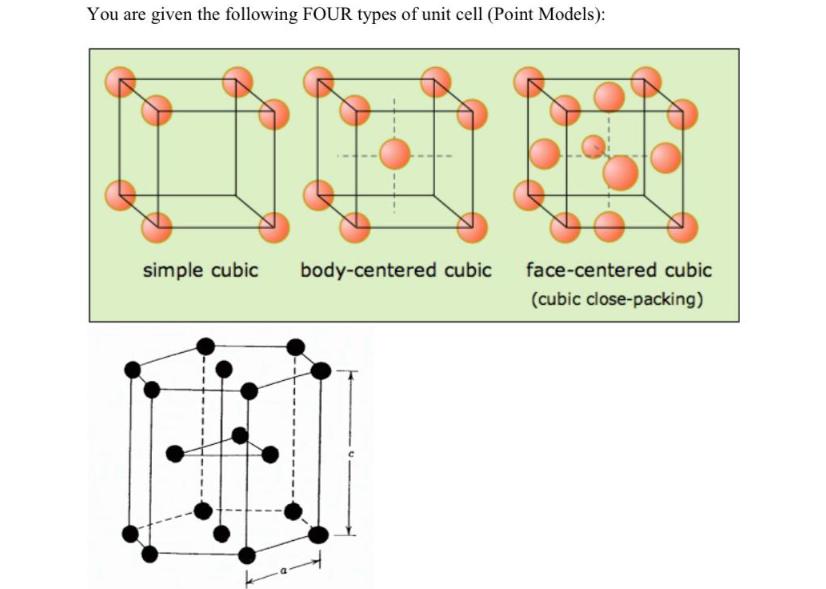
By overlying THREE two-dimensional layers of specifically packed atoms (around 20 atoms per layer), please demonstrate the 3-layers organization of i) body center cubic unit cells; ii) face-centered cubic and iii) Hexagonal Closed Pack unit cell [10 points] Please calculate the atomic packing factor of a Hexagonal Closed Pack (HCP) unit cell (Please show all the steps in your calculation) [25 points] You are given the following FOUR types of unit cell (Point Models): simple cubic body-centered cubic face-centered cubic (cubic close-packing) M By overlying THREE two-dimensional layers of specifically packed atoms (around 20 atoms per layer), please demonstrate the 3-layers organization of i) body center cubic unit cells; ii) face-centered cubic and iii) Hexagonal Closed Pack unit cell [10 points] Please calculate the atomic packing factor of a Hexagonal Closed Pack (HCP) unit cell (Please show all the steps in your calculation) [25 points] You are given the following FOUR types of unit cell (Point Models): simple cubic body-centered cubic face-centered cubic (cubic close-packing) M
Expert Answer:
Answer rating: 100% (QA)
The three unit cells are BodyCentered Cubic BCC Unit Cell Atoms are located at the corners of the cu... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The Calabrese Government Watchdog Group is considering a 4-year investment opportunity with the following cash flows: If Calabrese uses an annual discount rate of 8 percent, should it pursue the...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
1. What is Anthropology, Sociology, and Political Science in your own understanding? 2. What is the nature of Anthropology, Sociology, and Political Science? 3. How are they similar with one another?
-
1. The final simplex tableau for the dual of the linear programming problem of Exercise 3 is as follows. Give the solution to the problem and to its dual. 2. The final simplex tableau for the linear...
-
They complain about how quotas have risen over the year but not wages. Then, Dennis makes the revealing comment, I'm not sure that I could meet it even if I tried, and I'm sure as hell not going to...
-
A toroid carries a current \(I\) and has \(n\) circular windings per unit length measured along the inside edge of the windings. The radius of each circular winding is \(R_{\text {winding }}\), and...
-
Oceanwide Enterprises, Inc., is involved in building and operating cruise ships. Each ship is identified as a separate discrete job in the accounting records. At the end of 2010, Oceanwide correctly...
-
Automobile demand has fallen during a recent recession, and Ana has been laid off from her job on the assembly line. Rajiv recently lost his job as a waiter at a local restaurant. A recent increase...
-
Calculate the support reactions for this simply supported beam, a component of the bridge. Key Values A 15 kN B 20 KN C 3 kN/m X 1.5 m y 2.5 m R1 X A 1m B Y UDL = C R2 1m
-
If a letter of intent was signed which stated in part "make every reasonable effort to agree upon and have prepared as quickly as possible" a contract for the purchase of a car. but then the seller...
-
What is an example of a cost that is relevant in a long term decision, but not a short term decision?
-
(a) Show that every straight line in the Argand diagram can be represented by an equation of the form az + bz + c = 0 where a, b, c are (complex) constants. (Hint: Let z and 22 be two distinct point...
-
Nikola Corp. (NC) is trying to decide whether to lease or buy a new computer-assisted manufacturing system from Finland. Management has decided that it must use the system to remain competitive. The...
-
How many grams of CO are produced by CH4 in the following reaction by 16 g of CH4? CH4 + 202 CO + 2HO O 11264 g O 44 g O 22 g O 0.023 g
-
Use the below Employee turnover dataset Excel file to use R analysis Participants Job Satisfaction Work-Life Balance Career Management Employee turnover D6526 3.00 3.00 2.00 3.00 D6528 3.00 2.00 2.00...
-
TABLE 6 Present Value of an Annuity Due of $1 (1+ irx (1 + i) PVAD - n/i 1.0% 1.5% 2.0% 2.5% 3.0% 3.5% 4.0% 4.5% 5.0% 5.5% 6.0% 7.0% .0% 9.0% 10.0% 11.0% 12.0% 20.0% 1 1.00000 1.00000 1.00000 1.00000...
-
In the synthesis of the keto acid just given, the dicarboxylic acid decarboxylates in a specific way; it gives Explain. HO rather than HO
-
What is entropy? Why is entropy important in discussing the formation of solutions?
-
Write the formula for each molecular compound. a. Phosphorus trichloride b. Chlorine monoxide c. Disulfur tetrafluoride d. Phosphorus pentafluoride
-
How do solids, liquids, and gases differ?
-
Figure 7.26 shows a cart striking a spring. In Figure 7.26a, consider the motion of the cart along the direct path from the initial position \(x_{1}\), which is the position at which the cart makes...
-
Standing a few meters from a skateboard, a boarder begins running and then jumps onto the board. Make bar diagrams like the ones in Figure 7.32 to illustrate the energy conversions \((a)\) while he...
-
Two carts are about to collide head-on on a track. The inertia of cart 1 is greater than the inertia of cart 2, and the collision is elastic. The speed of cart 1 before the collision is higher than...

Study smarter with the SolutionInn App


