a) How many moles of CaBr2 would be consumed when reacted with 53.0 ml of a...
Fantastic news! We've Found the answer you've been seeking!
Question:
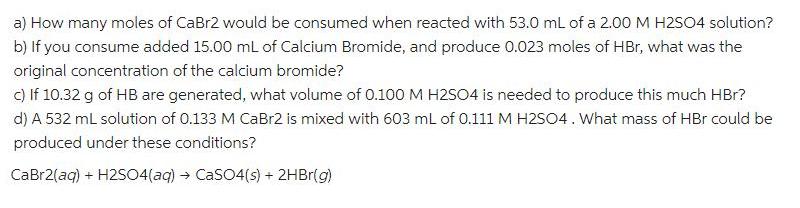
Transcribed Image Text:
a) How many moles of CaBr2 would be consumed when reacted with 53.0 ml of a 2.00 M H2S04 solution? b) If you consume added 15.00 mL of Calcium Bromide, and produce 0.023 moles of HBr, what was the original concentration of the calcium bromide? c) If 10.32 g of HB are generated, what volume of 0.100 M H2504 is needed to produce this much HBr? d) A 532 mL solution of 0.133 M CAB12 is mixed with 603 mL of 0.111 M H2SO4. What mass of HBr could be produced under these conditions? CaBr2(ag) + H2S04(aq) → CaSO4(s) + 2HBr(g) a) How many moles of CaBr2 would be consumed when reacted with 53.0 ml of a 2.00 M H2S04 solution? b) If you consume added 15.00 mL of Calcium Bromide, and produce 0.023 moles of HBr, what was the original concentration of the calcium bromide? c) If 10.32 g of HB are generated, what volume of 0.100 M H2504 is needed to produce this much HBr? d) A 532 mL solution of 0.133 M CAB12 is mixed with 603 mL of 0.111 M H2SO4. What mass of HBr could be produced under these conditions? CaBr2(ag) + H2S04(aq) → CaSO4(s) + 2HBr(g)
Expert Answer:
Answer rating: 100% (QA)
a mol of H2SO4 MV 2 53103 0106 mol of H2S04 ratio is 11 so mol of ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
You have 1.5 moles of tricycles. a. How many moles of seats do you have? b. How many moles of tires do you have? c. How could you use parts a and b as an analogy to teach a friend about the number of...
-
How many moles of O are needed to combine with 0.212 mole of C to form (a) CO (b) CO2?
-
How many moles of Al2O3 will be produced when 23.9 g of H2O are reacted according to this chemical equation? 2AlCl3 + 3H2O() ( Al2O3 + 6HCl(g)
-
Pick a method of accounting for bad debt. What do you like about it and why? give an example of a company where it makes sense to use that method? Why does it make sense? Also give an example of a...
-
On February 4, Campbell Company sells inventory to a customer for $6,000. Terms of the sale are 1/15, net 30. On February 10, the customer returns $500 of merchandise. The customer pays on February...
-
Would you prefer the use of bottom-up or top-down budgeting for project cost control? What are the advantages and disadvantages associated with each approach?
-
What is a bona fide occupational qualification (BFOQ)?
-
If we can get that new robot to combine with our other automated equipment, well have a complete flexible manufacturing system (FMS) in place in our Northridge plant, said Hal Swain, production...
-
Legacy issues $ 6 1 0 , 0 0 0 of 6 . 5 % , four - year bonds dated January 1 , 2 0 2 1 , that pay interest semiannually on June 3 0 and December 3 1 . They are issued at $ 5 4 0 , 9 8 1 when the...
-
The Dorton University president has asked the OSCM department to assign eight biology professors (A, B, C, D, E, F, G, and H) to eight offices (numbered 1 to 8 in the diagram) in the new biology...
-
Three point charges q are placed at the corners of an equilateral triangle. Another point charge -Q is placed at the centroid of the triangle. If the force on each of the charges q vanishes, then the...
-
Describe how the definition of marriage has evolved over the past 200 years.
-
Explain the principle of working of a thermocouple for temperature measurement.
-
Distinguish between prohibitions against marriage based on consanguinity and affinity and give an example of each.
-
Describe the significance of the Supreme Courts decision in Loving v. Virginia.
-
Explain the working of a manometer for pressure measurement.
-
Article Summary : read the article "Arbitration pros and cons" and summarize two (2) pros and two (2) cons https://www.nolo.com/legal-encyclopedia/arbitration-pros-cons-29807.html
-
Assume that your audit team has established the following parameters for the examination of ELM's sales transactions: LO G-3 Risk of incorrect acceptance...
-
Given that the reaction Cl 2 (g) + 2KBr(aq) 2KCl(aq) + Br 2 (aq) readily occurs, would you expect the reaction I 2 (s) +2KCl(aq) 2KI(aq) + Cl 2 (aq) to occur?
-
At what temperature does the rms speed of O2 molecules equal 400. m/s?
-
A 20.0-L vessel is filled with 1.00 mol of ammonia, NH3. What percent of ammonia dissociates to the elements if equilibrium is reached at 345oC? Use data from Appendix C and make any reasonable...
-
Using a financial calculator, solve for the unknowns in each of the following situations. a. On June 1, 2024, Holly Golightly purchases lakefront property from her neighbor, George Peppard, and...
-
Ed owns Oak Knoll Apartments. During the year, Fred, a tenant, moved to another state. Fred paid Ed \($1,000\) to cancel the two-year lease he had signed. Ed subsequently began renting the unit to...
-
In 2017, Harry and Mary purchased Series EE bonds, and in 2023 redeemed the bonds, receiving \($500\) of interest and \($1,500\) of principal. Their income from other sources totaled \($30,000.\)...

Study smarter with the SolutionInn App


