Calculate K at 298 K for the following reaction: 2 NO(g) + Cl(g) = 2 NOCI(g)...
Fantastic news! We've Found the answer you've been seeking!
Question:
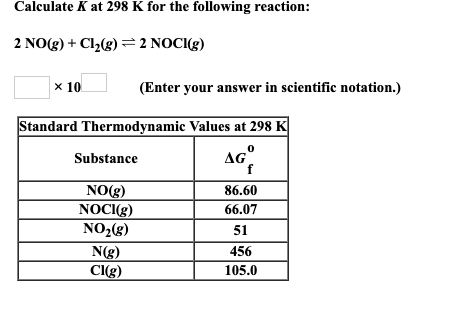
Transcribed Image Text:
Calculate K at 298 K for the following reaction: 2 NO(g) + Cl₂(g) = 2 NOCI(g) x 10 Standard Thermodynamic Values at 298 K AGº Substance NO(g) NOCI(g) NO₂(g) (Enter your answer in scientific notation.) N(g) Cl(g) 86.60 66.07 51 456 105.0 Calculate K at 298 K for the following reaction: 2 NO(g) + Cl₂(g) = 2 NOCI(g) x 10 Standard Thermodynamic Values at 298 K AGº Substance NO(g) NOCI(g) NO₂(g) (Enter your answer in scientific notation.) N(g) Cl(g) 86.60 66.07 51 456 105.0
Expert Answer:
Answer rating: 100% (QA)
Answer Ans 2NOg Cl 9 2 NOC 9 Aceron Auf product suf ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate K at 298 K for the following reaction: SrSO4(s)<--> Sr2+(aq) + SO42-(aq) SCIENTIFIC NOTATION PLEASE...thankyou . Comments : the answer K = 3.48 x 10^-7was wrong,it said HInt:For any...
-
Calculate the standard enthalpy change, Ho, for the following reaction at 25oC. Fe2O3(s) + 2Al(s) 2Fe(s) + Al2O3(s) What is the enthalpy change per mole of iron?
-
Calculate the standard enthalpy change, Ho, for the following reaction at 25oC. 3CaO(s) + 2Al(s) 3Ca(s) +Al2O3(s) What is the enthalpy change per mole of calcium?
-
The retail inventory method is used by The Bronx Company. Inventory, purchases, and sales data for 2016 are as follows: Using the retail method, calculate the Lower-of-Average-Cost or Market of the...
-
The Isabelle Corporation rents prom dresses in its stores across the southern United States. It has just issued a five-year, zero-coupon corporate bond at a price of $74. You have purchased this bond...
-
It's amazing how much difference there is in the way proposals are presented at two different firms," said John Woods to his assistant, Pete Madsen, as he pointed to the stack of capital investment...
-
Explain how the control variate technique is implemented.
-
Drew Company reports the following costs and expenses in May. InstructionsFrom the information, determine the total amount of:(a) Manufacturing overhead.(b) Product costs.(c) Period costs. Factory...
-
You are the COO managing the operations of a robo-advisor and you need to ensure that all the stock holdings in the portfolios under management are kept up to date. Given the following inputs in...
-
1. Why wasnt money an adequate remedy in this case? 2. What does Wilcox mean when he alleges that Shollmier engaged in collusion? 3. How could Wilcox have prevented the property from being sold below...
-
HARMONIZED SALES TAX (HST) LES MEUBLES D'ANTAN is a furniture retailer specializing in the sale of quality solid wood furniture. The company uses a periodic inventory system to record inputs and...
-
Discuss on what grounds an intervention order would be granted in South Australia and discuss the difference between criminal and civil legislation in relation to intervention orders. You are working...
-
Find the largest interval containing x = 0 that the Remainder Estimation Theorem allows over which f(x) = sin(3x) can be approximated by p(x) = 3x - decimal-place accuracy throughout the interval....
-
GBC Inc. experiences a loss of $100000 in 2022 and therefore cannot pay any dividends to shareholders. How will this development affect the value of shareholder's equity on the GBC Inc. Statement of...
-
Briefly explain the theory of Convergence relating to Economic Growth? How does Absolute Convergence differ from Conditional Convergence?
-
A client is unhappy with her former lawyer's account. She plans to complain to the Law Society that the lawyer has charged an excessive fee, and ask the Law Society to reduce the amount. a. Explain...
-
Edna Robson is a marketing manager for Blossom Cosmetics in Lethbridge, Alberta. She earns $45,000.00 annually and is paid on a semi-monthly basis. Edna's federal and provincial TD1 claim codes are...
-
What services are provided by the provincial and territorial governments?
-
Predict the sign of ÎSo for each of the following changes. a. b. AgCl(s) Ag+(aq) + Cl-(aq) c. 2H2(g) + O2(g) 2H2O(l) d. Na(s) + 1/2 Cl2(g) NaCl(s) e. HCl(g) H+(aq) + Cl2(aq) f. KBr(s) K+(aq) +...
-
Express the composition of each of the following com-pounds as the mass percent of its elements. a. Formaldehyde, CH 2 O b. Glucose, C 6 H 12 O 6 c. Acetic acid, HC 2 H 3 O 2 Considering your...
-
Identify the elements that correspond to the following atomic numbers. Label each as either a noble gas, a halogen, an alkali metal, an alkaline earth metal, a transition metal, a lanthanide metal,...
-
Fill in the blanks to make the following statements correct. a. Economists have designed____________ to better explain and predict the behaviour we observe in the world around us. b. A variable, such...
-
The following supply and demand schedules describe a hypothetical Canadian market for potash. a. What is the equilibrium price of potash? b. How much potash would actually be purchased if the price...
-
According to Statistics Canada, Canada's exports and imports of energy (combined totals of fossil fuels, hydro, and nuclear, all measured in petajoules) over a five-year period were as follows: a....

Study smarter with the SolutionInn App


