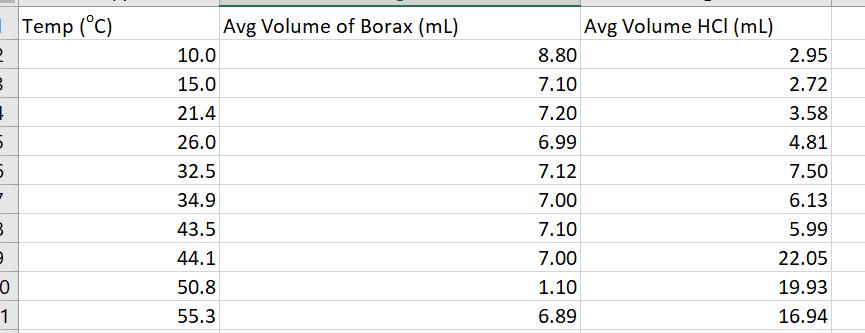
calculate ln Ksp 2 3 1 5 5 7 B 9 0 1 Temp (C) 10.0 15.0
Fantastic news! We've Found the answer you've been seeking!
Question:
calculate ln Ksp
Transcribed Image Text:
2 3 1 5 5 7 B 9 0 1 Temp (°C) 10.0 15.0 21.4 26.0 32.5 34.9 43.5 44.1 50.8 55.3 Avg Volume of Borax (ml) 8.80 7.10 7.20 6.99 7.12 7.00 7.10 7.00 1.10 6.89 Avg Volume HCI (mL) 2.95 2.72 3.58 4.81 7.50 6.13 5.99 22.05 19.93 16.94 2 3 1 5 5 7 B 9 0 1 Temp (°C) 10.0 15.0 21.4 26.0 32.5 34.9 43.5 44.1 50.8 55.3 Avg Volume of Borax (ml) 8.80 7.10 7.20 6.99 7.12 7.00 7.10 7.00 1.10 6.89 Avg Volume HCI (mL) 2.95 2.72 3.58 4.81 7.50 6.13 5.99 22.05 19.93 16.94
Expert Answer:
Answer rating: 100% (QA)
Ans Borate has the formula which is basicdianionic B 4 O 5 OH 4 2 if we add acid th... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Using the following data, calculate the value of Ksp for Ba(NO3)2, one of the least soluble of the common nitrate salts. Species Baa) NO ia Ba(NOs2s)- -561 kJ/mol -109 kJ/mol -797 k/mol
-
Consider the following galvanic cell: Calculate the Ksp value for Ag2SO4(s). Note that to obtain silver ions in the right compartment (the cathode compartment), excess solid Ag2SO4 was added and some...
-
Ksp for silver chloride at 25.0oC is 1.782 1010. At 35.0oC, Ksp is 4.159 1010. What are Ho and So for the reaction?
-
(a) Use a graphing utility to graph the curve given by (b) Describe the graph and confirm your result analytically. (c) Discuss the speed at which the curve is traced as t increases from -20 to 20. x...
-
A tobacco company advertises that the average nicotine content of its cigarettes is at most 14 milligrams. A consumer protection agency wants to determine whether the average nicotine content is in...
-
The assembly consists of two 10-mm diameter red brass C83400 copper rods AB and CD, a 15-mm diameter 304 stainless steel rod EF, and a rigid bar G. If the horizontal displacement of end F of rod EF...
-
It Wires aren't really ideal. The voltage drop across a currentcarrying wire can be significant unless the resistance of the wire is quite low. Suppose a \(50 \mathrm{ft}\) extension cord is being...
-
The BackPack Company produces a line of backpacks. The manager, Jill Nicholas, is interested in using a level aggregate plan. Inventories and back orders will be used to handle demand fluctuations....
-
(1) A = 3i - 4j + 6k, B = 4i + 6j - 2 k (a) What is the angle between there two vectors? (b) Show that RB=-BR
-
The finance director of RM plc is considering several investment projects and has collected the following information about them. Projects D and E are mutually exclusive. The capital available for...
-
164. A drug was studied in mice for its hepatotoxicity. The drug was injected into the mice at a dose of 10mg/kg & 20 mg/kg and biopsy from liver was taken. Hepatocytes appeared increased in size...
-
An article in the Economist magazine in 2012 observed: America can now borrow from the bond market at a cheaper rate than at any time in the history of the republic. Use the loanable funds model to...
-
An article in the New York Times in 2012 observed: Older Americans and other savers are just unintended casualties of policies aimed at other economic targets, particularly the policy making it...
-
In 2012, an article in the Economist magazine recommended to investors that if economic growth and inflation remained low in the United States, the investors should buy bonds. But if inflation...
-
You are the senior accountant for a business that regularly imports spare parts for a range of your products from overseas suppliers. You have been instructed by the CEO to look at ways you can save...
-
In which trading centers does 75 percent of all foreign exchange trading take place?
-
consumer risk in cereal when exporting to brazil and how to manage that risk
-
(8%) Problem 6: A student attaches a f= 3.5 kHz oscillator to one end of a metal rail of length L = 25 m. The student turns on the oscillator and uses a piezoelectric gauge at the other end to...
-
The enthalpy of vaporization of methanol is 35.27 k] mol-I at its normal boiling point of 64.1oC. Calculate (a) The entropy of vaporization of methanol at this temperature and (b) The entropy change...
-
Indicate on the phase diagram in Fig. 6.42 the feature that denotes incongruent melting. What is the composition of the eutectic mixture and at what temperature does it melt?
-
Explain the symmetry criteria that allow a molecule to be polar?
-
What do you understand by a bank lending policy in the context of a monetary policy?
-
A columnist in the Wall Street Journal argues: Whether youre a borrower or a saver, what matters isnt the nominal interest rate but the real, post-inflation rate of return. Do you agree? Briefly...
-
What is the relationship between the MPC and the multiplier? Would a larger value for the MPC tend to increase or decrease the stability of the economy? Briefly explain.

Study smarter with the SolutionInn App


