Naturally occurring lead exists as four stable isotopes: 204X with a mass of 203.973 amu (1.4806);...
Fantastic news! We've Found the answer you've been seeking!
Question:

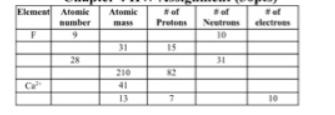
Transcribed Image Text:
Naturally occurring lead exists as four stable isotopes: 204X with a mass of 203.973 amu (1.4806); 206X 205.974 amu (23.60%); 207X 206.9759 amu (22.60%); and 20X 207.9766 amu (52.30%). Calculate the average atomic mass of X. 68.9257 amu is the mass of 60.4% of the atoms of an element with only two naturally occurring isotopes. The atomic mass of the other isotope is 70.9249 amu. Determine the average atomic mass of the element? Identify the element? Give the nuclear composition and isotopic notation for a, an atom containing 27 protons, 32 neutrons, and 27 electrons b. an atom containing 15 protons, 16 neutrons, and 15 electrons c. an atom containing 110 neutrons, 74 clectrons, and 74 protons d. an atom containing 92 electrons, 143 neutrons, and 92 protons Ekment of of ef Atomie Atomic Rumber Protons Neutrons electrees mass 10 15 28 31 210 Ca 13 10 Naturally occurring lead exists as four stable isotopes: 204X with a mass of 203.973 amu (1.4806); 206X 205.974 amu (23.60%); 207X 206.9759 amu (22.60%); and 20X 207.9766 amu (52.30%). Calculate the average atomic mass of X. 68.9257 amu is the mass of 60.4% of the atoms of an element with only two naturally occurring isotopes. The atomic mass of the other isotope is 70.9249 amu. Determine the average atomic mass of the element? Identify the element? Give the nuclear composition and isotopic notation for a, an atom containing 27 protons, 32 neutrons, and 27 electrons b. an atom containing 15 protons, 16 neutrons, and 15 electrons c. an atom containing 110 neutrons, 74 clectrons, and 74 protons d. an atom containing 92 electrons, 143 neutrons, and 92 protons Ekment of of ef Atomie Atomic Rumber Protons Neutrons electrees mass 10 15 28 31 210 Ca 13 10
Expert Answer:
Answer rating: 100% (QA)
Q1 Isotope abundance Atomic mass amu X204 1480 203973 X206 2360 205974 X207 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Protons and neutrons (together called nucleons) are held together in the nucleus of an atom by a force called the strong force. At very small separations, the strong force between two nucleons is...
-
Symbol Atomic # Protons Neutrons Atomic Mass Na 11 11 24 - 11 = 13 24 24-11: 24 Na 13
-
Protons with a kinetic energy of 2.00 MeV scatter off gold nuclei in a foil target. Each gold nucleus contains 79 protons. If both the incoming protons and the gold nuclei can be treated as point...
-
Solve the inequalities and show the solution sets on the real line. -2x > 4
-
The speed limit in a school zone is 40 km/h (about 25 mi/h). A driver traveling at this speed sees a child run onto the road 13 m ahead of his car. He applies the brakes, and the car decelerates at a...
-
Binary Star-Different Masses two stars, with masses M, and M2, are in circular orbits around their center of mass. The star with mass M, has an orbit of radius R the star with mass M2 has an orbit of...
-
Recall, there is some disincentive for parties to elect a trial de novo after receiving an adverse arbitration award. Further investigate CCP 1141.21 and determine the costs and fees that the party...
-
Would buying a car from a mechanic who works at a car dealership qualify as purchasing a car in the ordinary course of business? [Steele v Ellis, 961 F Supp 1458 (D Kan)]
-
Does APPLE issues stocks in foreign countries? If yes, are there any correlations in the stock price movements?
-
Figure 2 shows the support structure for a suspended apparatus in a laboratory. The apparatus is hung from the 1/4" A36 plate that is welded to the 3/8" plate using E70XX electrodes. Assuming that...
-
Question 40 T.L.C. Enterprises just revised its capital structure from a debt-equity ratio of 0.30 to a debt-equity ratio of 0.45. The firm's shareholders who prefer the old capital structure should:...
-
4. (Discrete-time queue) Suppose that a surgeon can perform m = 1 elective surgeries. Every day, K (a random variable) new cases are added to the waiting list, with its p.m.f. given by a P(K k),...
-
What measures have been taken to ensure the proper functioning of food commodity markets and their derivatives and facilitate timely access to market information, including on food reserves, to help...
-
Review how effectively you manage work schedules and patterns to maintain a positive group environment and identify changes that you could make to schedules and patterns ( In residential care for...
-
The Tornado Integrated Project Team (Tornado IPT) is part of the UK Ministry of Defence's (MoD's) Defence Equipment and Support (DE&S) organisation. It is responsible for the provision of logistical...
-
Find I such that 4(x,y) = x+3kxy 2 may be Harmonic and. Hind Its Conjugate Harmonic
-
Assume a bank is in the process of restructuring a $1 million loan. The following information is provided. The current interest rate of the loan is 8% annually and matures at the end of this year....
-
Factor and simplify, if possible. Check your result using a graphing calculator. 3 cot 2 + 6 cot + 3
-
An electrochemical cell is made by placing an iron electrode in 1.00 L of 0.15 M FeSO4 solution and a copper electrode in 1.00 L of 0.036 M CuSO4 solution. a. What is the initial voltage of this cell...
-
An antacid tablet weighing 0.853 g contained calcium carbonate as the active ingredient, in addition to an inert binder. When an acid solution weighing 56.519 g was added to the tablet, carbon...
-
Write the electron configuration and the Lewis symbol for Ca2+ and for S2.
-
True or false? Explain: modularity reduces complexity because A. It reduces the effect of incommensurate scaling. B. It helps control propagation of effects.
-
True or false? Explain: hierarchy reduces complexity because A. It reduces the size of individual modules. B. It cuts down on the number of interconnections between elements. C. It assembles a number...
-
If one created a graph of personal friendships, one would have a hierarchy. True or false?

Study smarter with the SolutionInn App


