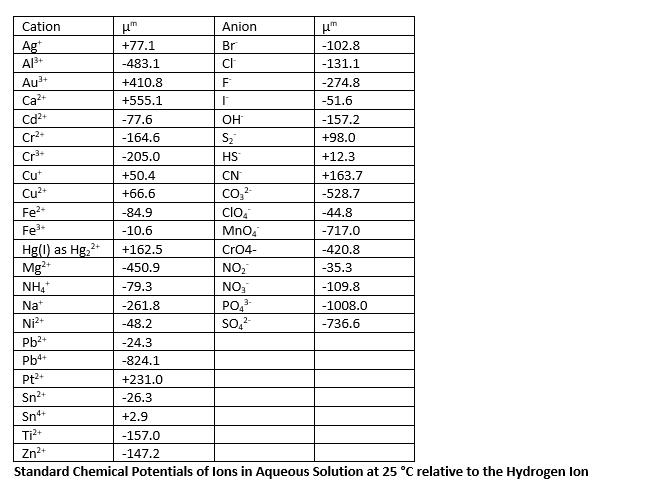
Calculate the half-cell potential of Ni in 0.1M NiCl2. The half-cell is represented as: NiNi2+, Cl- (0.1M).
Fantastic news! We've Found the answer you've been seeking!
Question:
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Posted Date:




